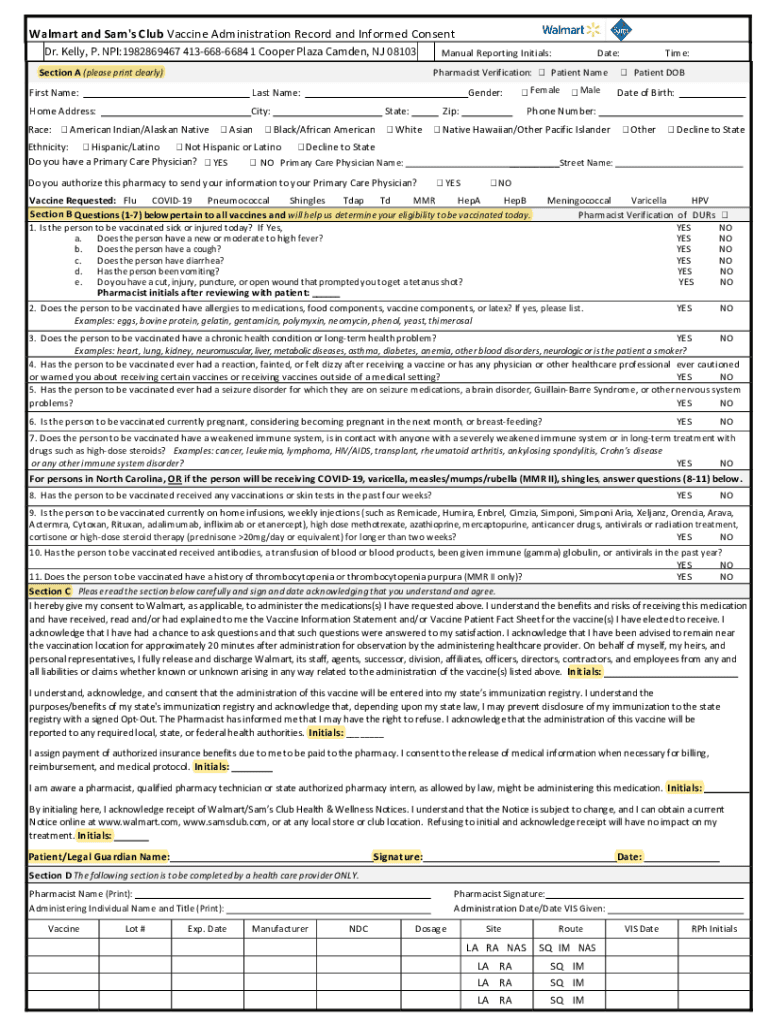
Definition and Purpose of the Vaccine Administration Consent Form
The vaccine administration consent form is a crucial document that provides patients with necessary information about the vaccine they are about to receive, including details on its purpose, benefits, and potential risks. It ensures that patients give informed consent before vaccination and outlines the responsibilities of both the patient and the administering healthcare professional.
- Purpose: To inform patients about the vaccine and obtain their consent.
- Legality: Functioning as a legally binding agreement that protects both patients and healthcare providers.
- Education: Helps to clarify patients' questions and concerns, fostering trust in the vaccination process.
This form typically consists of sections that require personal details, vaccination history, and consent declarations. Failing to complete the form could result in unintentional legal issues or miscommunication regarding a patient’s preferences.
Key Elements of Vaccine Administration Consent Form
A well-structured vaccine administration consent form includes several key components essential for clarity and completeness.
- Personal Information: This section collects data such as the patient's name, address, date of birth, and contact information.
- Vaccine Details: Clearly lists the type of vaccine, the dosage, and the intended benefits.
- Health History: Includes questions regarding allergies, previous vaccine reactions, and underlying medical conditions to assess eligibility.
- Consent Acknowledgment: A specific area where the patient confirms they understand the information provided and agree to receive the vaccine.
This comprehensive format ensures all necessary information is captured effectively, allowing healthcare providers to administer the vaccine safely and responsibly.
Completing the Vaccine Administration Consent Form
Filling out the vaccine administration consent form involves a systematic approach that ensures accuracy and completeness.
- Gather Personal Information: Fill out all fields accurately, including name, address, and relevant contact details.
- Review Vaccine Information: Carefully read the information provided about the vaccine, noting any potential side effects and benefits.
- Health History Disclosure: Complete the health history section truthfully to assist healthcare providers in making informed decisions.
- Consent Signature: Sign and date the form to affirm your consent and understanding of the vaccine being administered.
Providing clear and accurate information in the consent form helps prevent adverse reactions and ensures a smoother vaccination process.
Important Considerations for the Vaccine Administration Consent Form
When filling out the vaccine administration consent form, there are critical considerations to keep in mind.
- Legal Requirements: Understand that the form serves as a legal document that may be referred to in case of disputes or adverse events.
- Confidentiality: All information collected is subject to privacy laws, ensuring that patient data is handled securely.
- Right to Withdraw Consent: Patients retain the right to withdraw consent at any time prior to receiving the vaccine, which must be acknowledged on the form.
- Communication: Patients should feel encouraged to ask questions or express concerns regarding the vaccine and the form.
These considerations ensure that patients are fully aware of their rights and responsibilities associated with the vaccination process.
Risks and Benefits Acknowledgment in the Vaccine Administration Consent Form
A critical component of the vaccine administration consent form is the section that addresses the risks and benefits associated with vaccination.
- Benefits: Highlight the advantages of vaccination, including individual protection against disease, the potential for herd immunity, and overall public health benefits.
- Risks: Clearly outline possible side effects, ranging from mild reactions like soreness at the injection site to more severe but rare events.
- Informed Decision-Making: Inform patients that understanding these risks and benefits is essential for making an educated choice regarding their health.
This acknowledgment not only fulfills legal requirements but also strengthens the patient-provider relationship through transparency and cooperation.
Emergency Protocols Related to the Vaccine Administration Consent Form
Patients should be aware of the emergency protocols that may be implemented in case of adverse reactions to the vaccine post-administration.
- Immediate Reporting: Staff members must be trained to recognize and report any immediate adverse effects that require medical attention.
- Post-Vaccination Observation: Patients are often asked to remain in the clinic for a brief period after vaccination for monitoring purposes.
- Follow-Up Care: Information on whom to contact for follow-up questions, medical concerns, or reporting adverse events should be included on the form.
Understanding these protocols reassures patients of the safety measures in place during and after the vaccination process.