Definition and Meaning
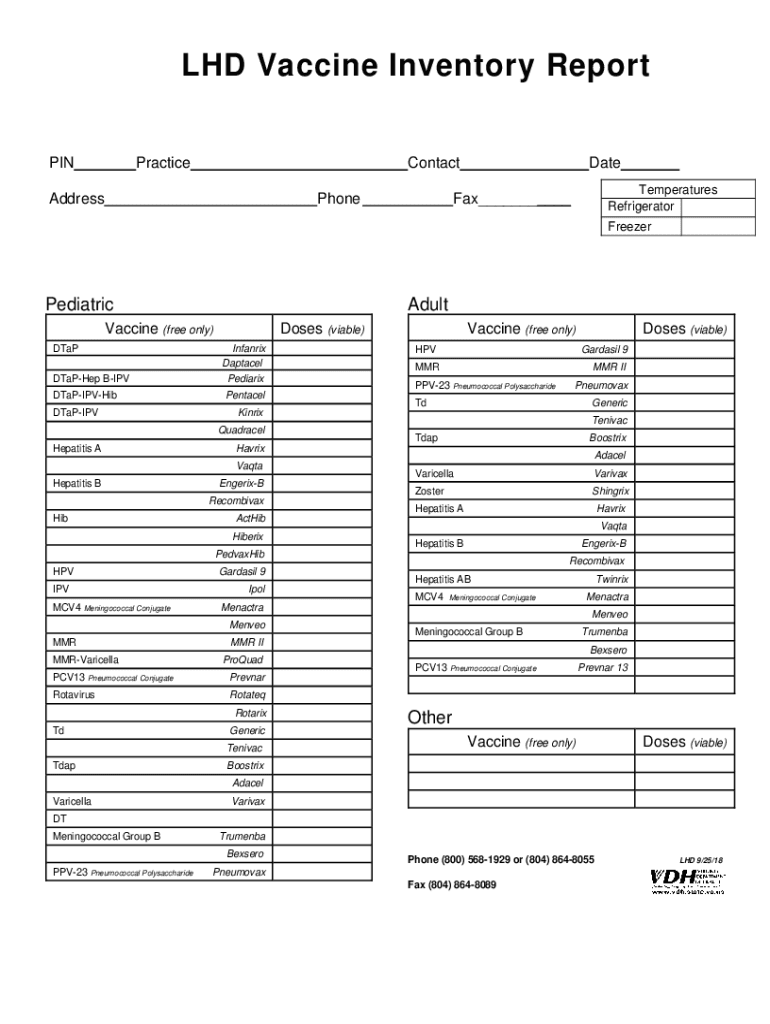
The "2018 VA LHD Vaccine Inventory Report" is a comprehensive document used by Local Health Departments (LHD) in Virginia to track vaccine inventories. Its primary purpose is to catalog all available vaccines, detailing their types, quantities, and storage conditions. This report ensures that each health department maintains accurate stock records, which is crucial for efficient public health management and distribution.
Key Components
- Vaccine Types and Quantities: The report provides a list of all vaccines available at the LHD, categorized by type and age group, which helps in planning immunization campaigns.
- Storage Details: It includes information on where each vaccine is stored, specifying whether they are kept in refrigerators or freezers, ensuring proper preservation conditions.
- Contact Information: For any inquiries, the report lists addresses, telephone numbers, and contact persons for each LHD, facilitating smooth communication channels.
How to Use the 2018 VA LHD Vaccine Inventory Report
This report serves as both a documentation tool and a reference point for health departments and policymakers. Users should regularly update the inventory to reflect any changes in stock levels due to vaccine administration or new deliveries.
Procedures for Effective Use
- Regular Updates: Ensure consistent documentation of all changes in vaccine quantities to keep the inventory accurate.
- Cross-checking Records: Periodically verify physical stocks against the inventory report to prevent discrepancies.
- Audit Preparations: Use the report as a primary source during internal and external audits to substantiate vaccine handling and storage practices.
Steps to Complete the 2018 VA LHD Vaccine Inventory Report
Filling out the report accurately involves several critical steps, ensuring all relevant data is captured.
- Gather Initial Data: Collect comprehensive information on current stock levels, including vaccine types and quantities.
- Identify Storage Locations: Document storage conditions for each vaccine type categorized either under refrigeration or freezing.
- Enter Contact Details: Fill in primary contact information for the LHD to facilitate communication.
- Review for Accuracy: Conduct a thorough review of the entered data to confirm its correctness before finalizing the report.
Important Terms Related to the 2018 VA LHD Vaccine Inventory Report
Understanding specific terminology is vital for accurately interpreting and using the vaccine inventory report.
- Cold Chain Management: Refers to the process of maintaining optimal temperature conditions for vaccines from manufacture through distribution to administration.
- Batch Number: A unique identifier for vaccine lots, essential for tracking in cases of recalls or adverse events.
- Expiry Date: Indicates the last date the vaccine can be used safely and effectively, crucial for planning vaccine usage.
Legal Use of the 2018 VA LHD Vaccine Inventory Report
This report holds significant legal implications for public health management, ensuring health departments comply with federal and state regulations regarding vaccine storage and distribution.
Compliance and Regulations
- Regulatory Requirements: LHDs must use the report to demonstrate adherence to vaccine storage and inventory management practices as required by the Centers for Disease Control and Prevention (CDC).
- Audit Documentation: The report can serve as evidence in audits, confirming the proper handling and documentation of vaccines.
Who Typically Uses the 2018 VA LHD Vaccine Inventory Report
The primary users of this report include various local health department officials and public health professionals involved in vaccine administration and inventory management.


Key Users
- Public Health Administrators: Use the report to track vaccine availability and storage practices.
- Vaccine Coordinators: Distribute vaccines based on inventory data to clinics and mobile teams.
- Healthcare Providers: Reference the report for planning immunization clinics and managing patient appointments effectively.
Key Elements of the 2018 VA LHD Vaccine Inventory Report
Understanding its critical components is essential for effectively managing vaccine inventories.
Major Components
- Detailed Vaccine Listings: Lists every available vaccine's type, quantity, and storage requirements.
- Regular Updates Section: Allows for updating inventory changes to ensure sustained accuracy over time.
- Storage Specifications: Details regarding refrigerator and freezer storage to ensure compliance with best practices.
State-Specific Rules for the 2018 VA LHD Vaccine Inventory Report
Certain state-specific regulations affect how Virginia's LHDs use the vaccine inventory report, ensuring adherence to local public health guidelines.
Virginia-Specific Guidelines
- State Department of Health Protocols: Outlines processes for maintaining vaccine stock levels to prevent shortages or oversupply.
- Reporting Compliance: Ensures that all vaccine inventory reports align with the Virginia Department of Health's mandates and are submitted on time for evaluation and planning.