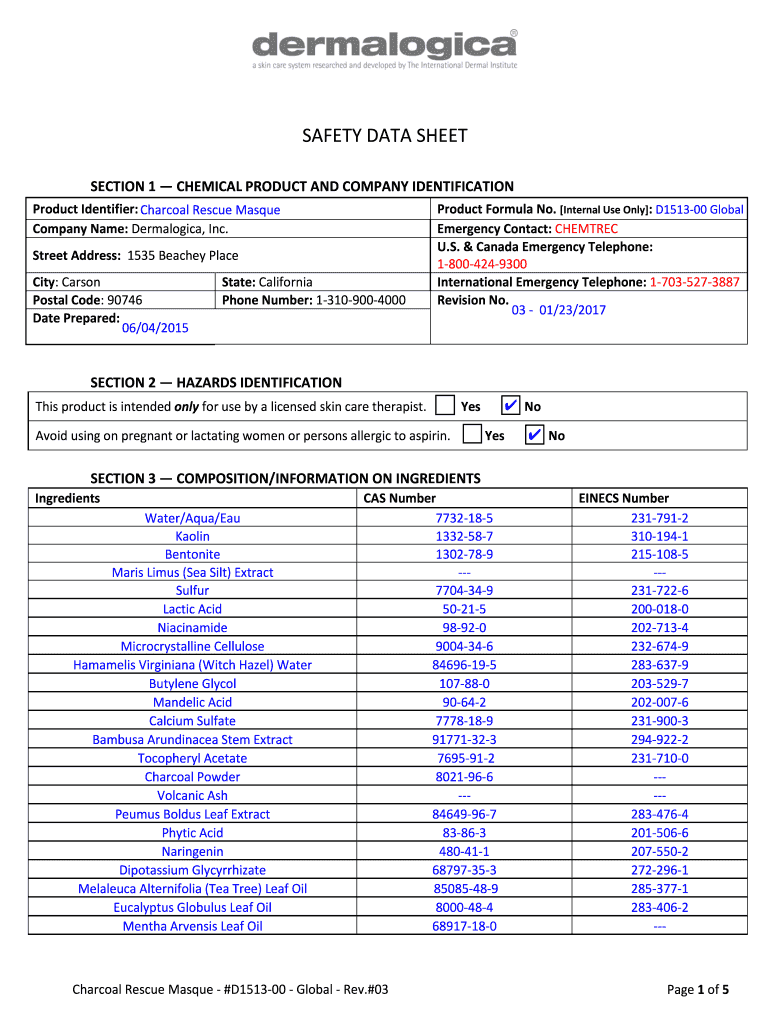
Understanding Dermalogica SDS Sheets
Dermalogica SDS Sheets, or Safety Data Sheets, are critical documents providing comprehensive information about the safe handling, transport, and use of specific cosmetic products produced by Dermalogica, Inc. These documents are essential for ensuring the safety of those who use or handle the products, including Charcoal Rescue Masque, Skin Hydrating Masque, and Skin Refining Masque. They cover a wide range of safety and regulatory information, which is necessary for adhering to U.S. legal standards in handling these cosmetics.
Key Elements of Dermalogica SDS Sheets
The Dermalogica SDS Sheets include 16 standardized sections that provide thorough details about each product:
- Product Identification: Describes the product name, brand, and appropriate uses.
- Hazard Identification: Detailed information on potential hazards associated with the product.
- Composition/Information on Ingredients: A breakdown of the chemical components present in the product.
- First Aid Measures: Instructions on how to respond to exposure to the product.
- Fire Fighting Procedures: Recommendations on how to safely extinguish a fire involving the product.
- Handling and Storage Guidelines: Best practices for storing and handling the product safely.
- Exposure Controls: Information on protective measures and exposure limits.
- Physical and Chemical Properties: Describes the product's appearance, odor, and other chemical properties.
- Stability and Reactivity: Details any chemical stability issues and reactivity with other substances.
- Toxicological Information: Potential toxic effects based on composition and usage.
- Ecological Impact: The product's effect on the environment.
- Disposal Considerations: Best practices for safe and lawful disposal of the product.
- Transport Information: Guidance for safely transporting the product.
- Regulatory Details: Information on regulatory compliance and standards.
- Disclaimers: Legal notifications regarding the information provided on the SDS.
How to Obtain Dermalogica SDS Sheets
To ensure proper handling and safety, it is crucial to access the SDS sheets before using or handling Dermalogica products. Typically, these sheets can be requested directly from Dermalogica's website or customer service. They are also often available via professional beauty suppliers or distributors who sell Dermalogica products. In some cases, the sheets can be found through industry databases dedicated to chemical safety.
Who Typically Uses Dermalogica SDS Sheets
The primary users of Dermalogica SDS Sheets are licensed skincare therapists and beauty industry professionals. These individuals rely on the comprehensive safety and regulatory information provided to ensure that products are used safely and in accordance with regulatory standards. In addition, safety officers in beauty salons, distributors, and regulatory agencies may also use these documents to inspect or verify compliance with safety standards.
Legal Use of Dermalogica SDS Sheets
SDS sheets are legally required by the Occupational Safety and Health Administration (OSHA) in the United States for products containing chemicals. They serve as a legal document for companies to demonstrate compliance with health and safety regulations. Therefore, ensuring the correct and up-to-date SDS is maintained and accessible could be imperative in the case of regulatory inspections or audits.
Importing Dermalogica SDS Sheets into Document Workflow
For institutions and business entities using platforms like DocHub for document handling, importing SDS sheets directly into the system ensures efficient access and sharing. This method prevents the loss of crucial safety information and makes it easier to distribute updated documents across teams.
- Use DocHub to upload SDS files directly from your computer or connected cloud services.
- Annotate important safety sections with the provided tools, such as highlighting crucial handling procedures.
- Set document access permissions to ensure authorized personnel have easy retrieval while maintaining sensitive data security.
Examples of Using Dermalogica SDS Sheets
One practical scenario involves a skincare therapist preparing for a treatment session. Before using any product, the therapist refers to its SDS to verify its safe application process and any personal protective equipment required. Another example is a distributor ensuring that all hazardous material shipments comply with transit regulations, referencing SDS sheets for accurate hazard communication.
Software Accessibility for Dermalogica SDS Sheets
Incorporating SDS sheets into digital workflows requires compatibility with document management software which can handle various file types, including PDFs. Platforms like DocHub ensure seamless integration by supporting a wide range of file formats, thus facilitating easy management, annotation, and collaboration on SDS documents across digital platforms without format conversion issues.
Important Terms Related to Dermalogica SDS Sheets
Understanding specific terms used in SDS sheets enhances comprehension and compliance:
- CAS Number: A unique identifier assigned to chemical substances.
- PPE (Personal Protective Equipment): Gear that individuals should wear to protect against product hazards.
- TLV (Threshold Limit Value): The level to which a worker can be exposed without adverse effects.
- MSDS (Material Safety Data Sheet): The previous version and format of current SDS.
These term definitions are crucial for interpreting safety recommendations accurately and ensuring compliance with legal and safety standards in professional environments.