Definition & Meaning
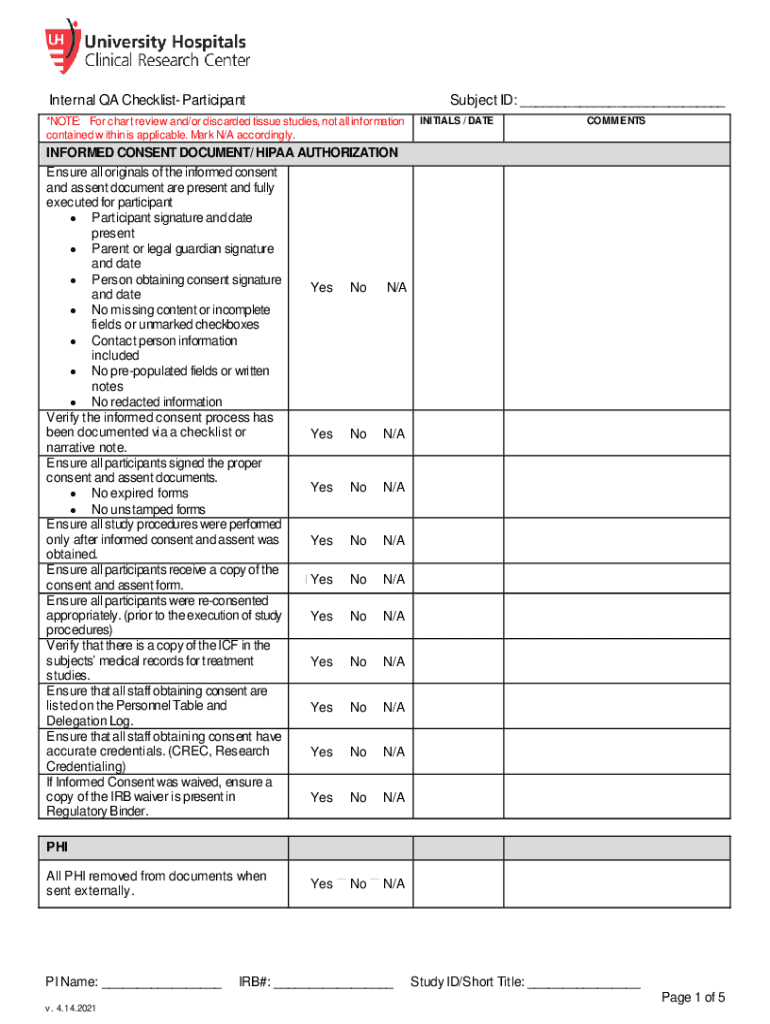
The "Internal QA Checklist - Regulatory Study ID: IRB#: PI Name:" is an essential document for maintaining compliance within research studies. It provides a structured approach to ensure that all research protocols meet required standards. The checklist is instrumental in verifying informed consent, eligibility, safety reporting, accountability for test articles, specimen handling, data collection, and financial documentation. By meticulously following this checklist, researchers can assure adherence to guidelines, safeguarding participant rights and data privacy, and maintaining the integrity of the study.
Key Elements of the Checklist
- Informed Consent: Ensures that participants are fully informed about the study's scope, risks, and benefits.
- Eligibility Criteria: Verifies that all participants meet the necessary criteria to partake in the study.
- Safety Reporting: Monitors and documents any adverse effects or safety concerns.
- Test Article Accountability: Tracks the usage and handling of any test articles used in the study.
- Specimen Handling: Ensures proper handling and storage of biological specimens.
- Data Collection and Financial Documentation: Confirms the accuracy and completeness of data and financial records related to the study.
How to Use the Internal QA Checklist
To effectively utilize the Internal QA Checklist, start by thoroughly reviewing each section to understand the parameters it covers. Here's a step-by-step guide:
- Prepare the Checklist: Download or access the checklist relevant to your study.
- Review Study Protocols: Align checklist items with your study protocols to ensure comprehensiveness.
- Assess Compliance: Systematically check each item to verify compliance.
- Document Findings: Record any deviations or compliance issues.
- Implement Corrections: Address and rectify identified issues promptly.
Practical Examples
- Case Study 1: In a clinical drug trial, researchers used the checklist to monitor patient eligibility and the informed consent process, ensuring that participants were adequately informed and met all criteria.
- Case Study 2: A research team applied the checklist to track specimen handling, maintaining sample integrity throughout the study duration.
Why You Need the Internal QA Checklist
The primary function of the Internal QA Checklist is to ensure compliance and quality assurance in research studies. Its benefits include:
- Risk Mitigation: Identifies potential issues early, reducing the risk of non-compliance.
- Standardization: Provides a uniform process to maintain consistency across various study protocols.
- Regulatory Compliance: Ensures adherence to institutional review board (IRB) and other regulatory requirements, such as the ESIGN Act for electronically signed documents.
- Participant Safety and Trust: Upholds ethical standards, fostering participant trust by protecting their rights and data.
Who Typically Uses the Checklist
- Principal Investigators (PIs): Primarily responsible for ensuring the study complies with all regulatory and ethical standards.
- Research Coordinators: Assist in implementing study protocols and completing the checklist.
- IRB Members: Use the checklist to audit and review study compliance.
- Regulatory Affairs Specialists: Ensure all compliance measures align with regulatory requirements.


Key Elements of the Internal QA Checklist
Understanding the components of the Internal QA Checklist is critical for proper application:
- Protocol Adherence: Verifies that study procedures align with the approved research protocol.
- Data Integrity: Confirms that all collected data is accurate and securely stored.
- Auditing and Monitoring: Ensures all research activities undergo regular audits and monitoring.
Legal Use of the Internal QA Checklist
The checklist plays a vital role in maintaining legal compliance throughout a research study. Ensuring adherence to the checklist can prevent legal implications related to:
- Informed Consent: Non-compliance can lead to ethical violations.
- Patient Safety: Neglecting safety protocols may result in regulatory infractions.
- Data Security: Mismanagement of data collection and storage may breach privacy laws.
Legal Context
- U.S. Regulations: Aligns with federal regulations, including the Common Rule and HIPAA.
- IRB Requirements: Ensures compliance with institutional commitments to ethical research conduct.
Steps to Complete the Internal QA Checklist
A structured approach is necessary for thorough completion:
- Initial Review: Familiarize yourself with the checklist items.
- Assign Responsibilities: Designate tasks to suitable team members.
- Conduct a Walkthrough: Go through each checklist item with relevant stakeholders.
- Document Evidence: Capture and store evidence of compliance for future reference.
- Submit Findings: Ensure all checklist items are validated and results documented.
- Follow-up Actions: Implement corrective actions for any issues found.
Important Terms Related to the Checklist
- IRB (Institutional Review Board): A committee that reviews research to ensure ethical standards.
- PI (Principal Investigator): The individual responsible for overseeing the conduct of the research study.
- Informed Consent: The process of obtaining voluntary participation from study subjects after informing them about the study details.
- Compliance: Adherence to specified standards and protocols.
By understanding and implementing the Internal QA Checklist thoroughly, research teams can ensure their studies meet essential regulatory standards and maintain high-quality research conduct.