Definition & Meaning
The Adverse Event Form is a structured document used primarily in clinical research and medical settings to record adverse events experienced by participants during a study or treatment regimen. An adverse event is any undesirable experience associated with the use of a medical product or procedure. This form serves as a critical tool for ensuring patient safety and regulatory compliance.
Adverse Event Forms facilitate the documentation and analysis of incidents that may impact the safety and efficacy of a treatment. The data collected helps in understanding the frequency, severity, and possible causation of these events, thereby contributing to ongoing research and patient safety measures.
Key Elements of the Adverse Event Form
Adverse Event Forms typically include several essential components designed to provide comprehensive information about each incident:
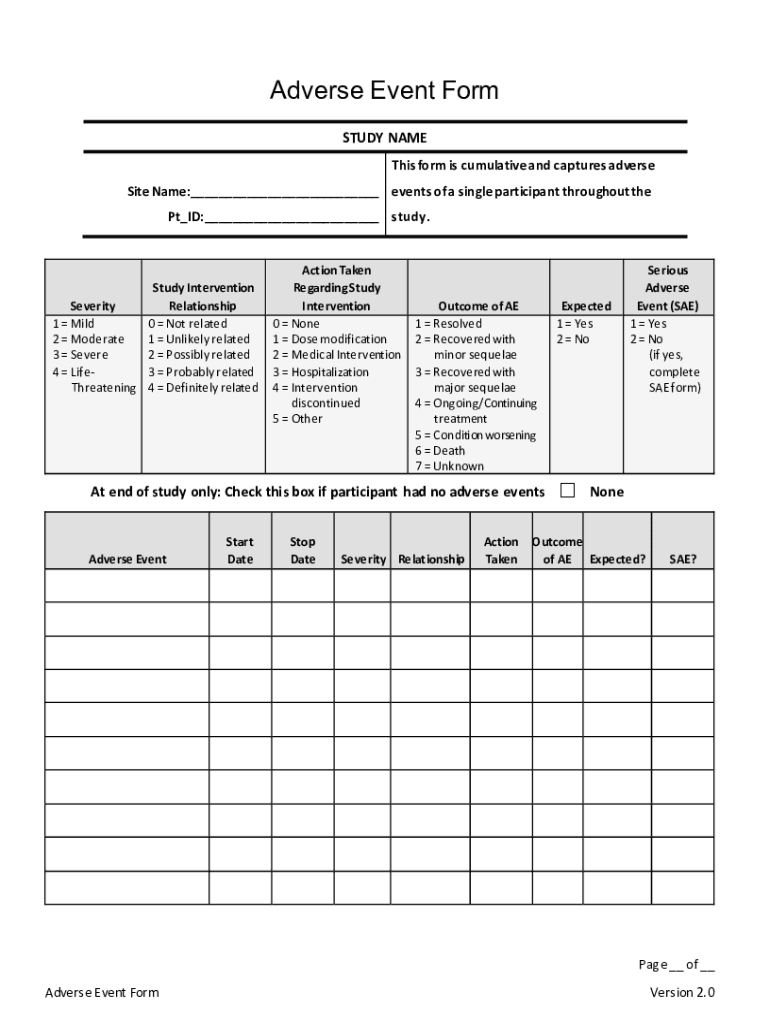
- Participant Information: Includes demographics such as age, sex, and participant ID, essential for correlating responses with specific sub-groups.
- Study Details: Refers to the study name, protocol number, and site location, which help in tracking and identifying the event within the broader research framework.
- Event Description: A detailed account of the adverse event, including date and time of occurrence, and a narrative section for additional observations.
- Severity Assessment: Rates the severity of the event from mild to severe, aiding in risk assessment and management.
- Causality Assessment: Determines the likelihood that the adverse event is related to the intervention rather than other factors.
- Outcome: Reports the resolution status of the adverse event, such as recovered, ongoing, or fatal.
Steps to Complete the Adverse Event Form
To accurately complete the Adverse Event Form, follow these steps:
- Gather Participant Information: Ensure that all identifying information is accurate and up-to-date.
- Document the Event: Provide a clear and detailed description of the adverse event, noting timing and circumstances.
- Assess Severity and Causality: Use standardized scales to evaluate the seriousness of the event and its potential connection to the study intervention.
- Record the Outcome: As the event resolves, document any treatments administered and the participant's response.
- Review and Submit: Ensure all sections are completed and reviewed by a qualified professional before submission to the relevant monitoring boards.
Legal Use of the Adverse Event Form
The Adverse Event Form is governed by stringent legal and ethical standards to protect participants and ensure data integrity:
- Compliance with Regulations: Adhere to FDA guidelines and other regulatory bodies, ensuring that adverse event reporting aligns with legal requirements.
- Confidentiality: Maintain participant privacy in accordance with HIPAA and other applicable privacy laws.
- Documentation: Ensure all events are documented truthfully and submitted timely to relevant authorities and within established reporting timelines.
Important Terms Related to the Adverse Event Form
Several key terms are critical for understanding and appropriately utilizing the Adverse Event Form:
- AE (Adverse Event): Any untoward medical occurrence in a study participant.
- SAE (Serious Adverse Event): Events leading to significant consequences such as hospitalization or death.
- Unexpected Adverse Event: An event that is not consistent with prior observations or documented risks.
Examples of Using the Adverse Event Form
Understanding real-world applications of the Adverse Event Form helps in appreciating its importance and versatility:
- Clinical Trials: Used extensively in documenting any participant reactions during drug testing phases.
- Vaccination Programs: Critical for ongoing assessment of vaccine safety in broader public health settings.
- Post-Market Surveillance: Manufacturers use these forms to monitor adverse events post-market release to ensure long-term product safety.
Digital vs. Paper Version
Both digital and paper versions of the Adverse Event Form have distinct advantages:
- Digital Forms: Allow for real-time data collection and integration into electronic health records, enhancing accessibility and speed.
- Paper Forms: Useful in settings with limited digital infrastructure, ensuring documentation is still accurately captured and stored.
Consider selecting the format that best suits your technological environment and regulatory requirements.
Software Compatibility
The integration of the Adverse Event Form with various software tools can streamline documentation processes:
- DocHub & Google Workspace: Facilitates online document creation, sharing, and signing securely.
- Specialized EDC Systems: Platforms like REDCap and Medidata enhance comprehensive data entry and monitoring in clinical trials.
- Standard Office Applications: Basic software like Microsoft Word or Excel can be used for form creation and data management.
Form Submission Methods
Different methods are available for submitting the Adverse Event Form, each catering to specific institutional needs:
- Online Submission: Preferred in settings with reliable internet infrastructure, allowing for quick and secure reporting.
- Mail-In Option: Utilized in settings that require physical documentation or archival purposes.
- In-Person Submission: Allows for direct communication and clarification with supervising bodies during submission.
Choosing the appropriate submission method is crucial for maintaining compliance and ensuring timely communication with regulatory authorities.