Definition and Purpose of Changes in Pharmacy Law
The "Changes in Pharmacy Law - California State Board of Pharmacy" document outlines updates to regulations impacting pharmacies within California. These modifications affect compliance requirements, operational procedures, and professional responsibilities. The update ensures that pharmacy practices align with evolving public health standards and technology. Understanding these changes is crucial for pharmacy operators, pharmacists, and stakeholders to maintain legal compliance and refine service delivery.
Key Elements of the Pharmacy Law Changes
Changes typically encompass adjustments in licensing procedures, record-keeping mandates, and the scope of pharmacist responsibilities. Elements often include:
- Amendments to pharmacist qualifications or continuing education requirements.
- Updated protocols for prescription management and patient confidentiality.
- Regulations surrounding the distribution of controlled substances.
Each update is formulated to enhance patient safety, streamline pharmacy operations, and address emerging issues such as telehealth services and remote dispensing practices.
Steps to Implement the Law Updates
To effectively incorporate the changes into daily operations:
- Review the Law: Thoroughly read the new regulations as published by the California State Board of Pharmacy.
- Assess Impact: Determine how the changes affect current policies and procedures.
- Train Staff: Conduct comprehensive training sessions to familiarize all pharmacy staff with new protocols and requirements.
- Update Documentation: Revise all relevant documentation, including standard operating procedures (SOPs) and patient consent forms.
- Audit Compliance: Regularly review pharmacy operations to ensure adherence to updated laws.
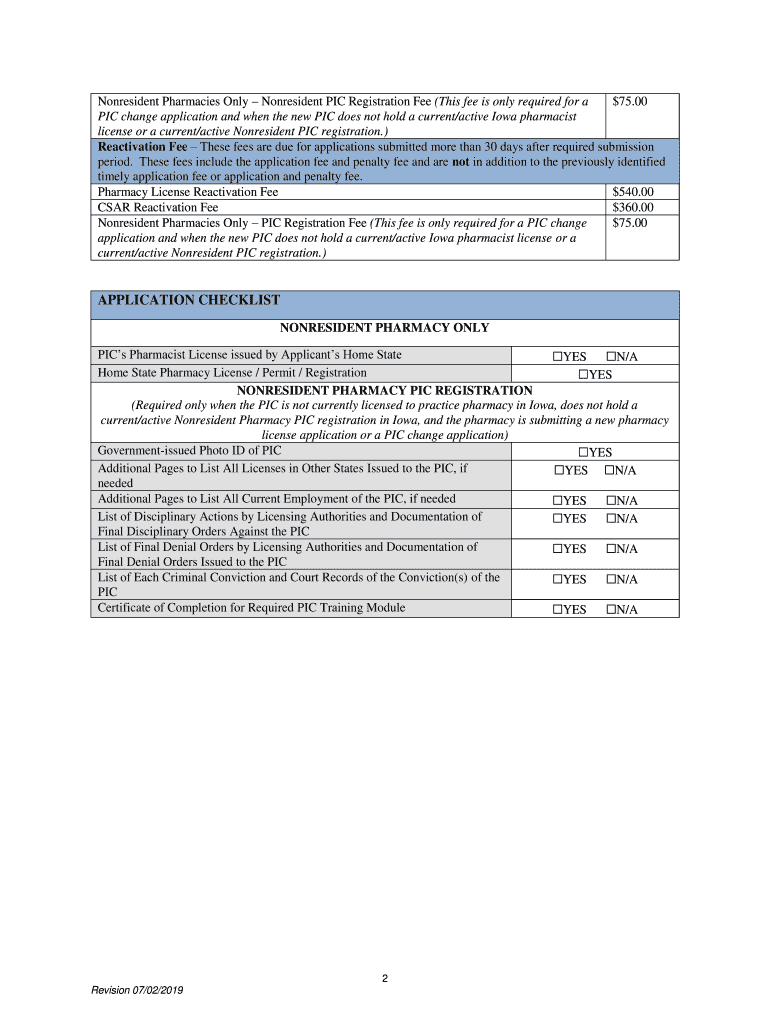
Required Documentation and Filing
Pharmacies are required to maintain specific documents to ensure compliance with updated laws. These often include:
- Revised pharmacy licenses or permits.
- Updated records of employee credentials and continuing education.
- Documentation of prescription handling and patient interactions in accordance with new laws.
Failure to maintain accurate and up-to-date documentation may result in legal penalties or operational disruptions.
State-Specific Rules and Variations
California's pharmacy regulations might differ from those in other states. Key distinctions often include:
- Unique licensure processes for pharmacists and pharmacy technicians.
- Special guidelines for compounding pharmacies in relation to the mixture and distribution of medications.
- Variations in patient consultation obligations, particularly concerning new prescription issuance.
Understanding these state-specific rules is essential for pharmacists operating near state borders or those involved in multi-state pharmacy operations.
Legal Implications for Non-Compliance
Non-compliance with the "Changes in Pharmacy Law" can result in serious legal consequences. Potential penalties include:
- Fines and penalties imposed by the California State Board of Pharmacy.
- Revocation or suspension of pharmacy licenses.
- Legal action from affected parties, particularly if patient safety is compromised.
Thus, strict adherence to updated laws is vital to avoid legal repercussions and protect the pharmacy's reputation.
Examples and Case Studies
Case studies often demonstrate how pharmacies successfully adapted to legal changes:
- A local pharmacy enhanced its prescription verification process following updates in patient privacy rules, resulting in increased customer trust.
- Chains implemented electronic tracking systems to comply with new record-keeping mandates, improving accuracy and compliance.
Such examples illustrate practical applications of legal updates and highlight potential benefits of adherence.
Who Typically Uses the Law Update Information
The information contained in the "Changes in Pharmacy Law" is of primary importance to:
- Pharmacists and Pharmacy Technicians: For operational changes and continuing education requirements.
- Pharmacy Owners and Managers: To implement necessary alterations in business practices.
- Healthcare Professionals: Ensuring collaborative compliance, particularly in integrative health practices involving pharmacists.
These stakeholders utilize the document to maintain compliance and ensure optimal patient care.


Form Submission Methods and Approval Timeline
For official submissions related to changes in pharmacy law, the California State Board of Pharmacy typically offers:
- Online Submission: Facilitated through the Board's official portal for rapid processing.
- Mail or In-Person Submissions: While less common, these may be required for specific forms or documentation that necessitate original signatures.
Turnaround times for approval can vary, with online applications generally processed more swiftly due to digital integration.