Definition & Meaning
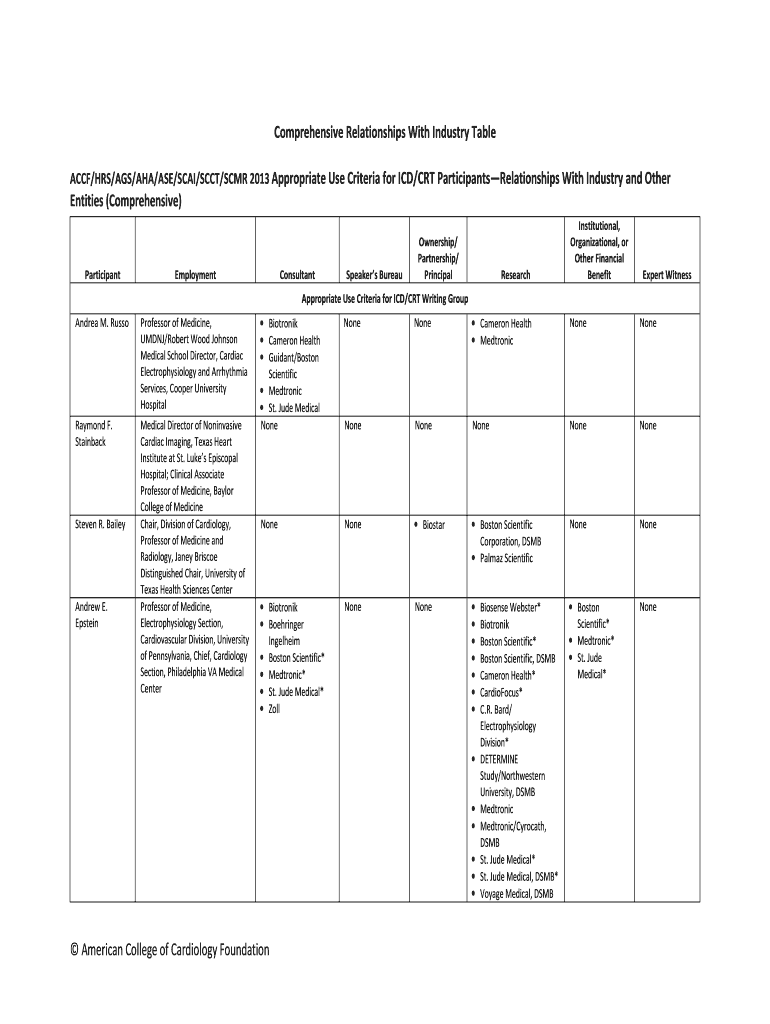
The Comprehensive Relationships With Industry Table ACCFHRS serves as a standardized framework aimed at clarifying the relationships between medical professionals and industries. This table is part of the documentation for the 2013 Appropriate Use Criteria for ICD/CRT and compiles critical information about the affiliations of participants, detailing aspects such as employment, consultancy roles, and financial interests. The goal is to provide transparency and prevent conflicts of interest in medical recommendations and industry partnerships.
How to Use the Comprehensive Relationships With Industry Table ACCFHRS
To effectively utilize this table, first, understand the categories outlined within it. Each row corresponds to a different participant and delineates their affiliations with industries or other entities, whether through employment or consultancy. The categorization of relationships – whether significant or modest – is based on ownership stakes and income levels. Review each section carefully, ensuring a comprehensive understanding of how each participant's affiliations might impact their contributions to the medical criteria.
Step-by-step Use
- Identify Participants: Locate each participant's name and role.
- Review Employment Status: Assess any direct employment with industry entities.
- Consulting Relationships: Check for consulting roles, including the scope and compensation.
- Research Affiliations: Observe research-related partnerships and funding.
- Financial Benefits: Evaluate the financial benefits received, focusing on equity ownership and compensation tiers.
Steps to Complete the Comprehensive Relationships With Industry Table ACCFHRS
Completing the table involves compiling and organizing data that captures various affiliations accurately. Start by collecting comprehensive participant information — their organizational roles, consulting services, research ties, and any financial benefits from industry relationships.
Compilation Process
- Collect Data: Gather data from the participants’ disclosures and financial documents.
- Verify Information: Cross-check all data for accuracy.
- Categorize Relationships: Classify each relationship as significant or modest, according to predefined criteria.
- Populate the Table: Enter the verified data into the appropriate sections of the table.
- Review for Accuracy: Conduct a thorough review to ensure no omissions or errors.
Key Elements of the Comprehensive Relationships With Industry Table ACCFHRS
The table includes several crucial elements necessary for conveying detailed information about industry relationships:
- Employment: Lists the organization and role of each participant.
- Consulting Roles: Details services rendered and any compensation received.
- Research Affiliations: Information on collaborative research and any funding or support.
- Financial Interests: Breakdown of financial interests, including stock ownership and payment details.
- Transparency: Notes on how participants maintain transparency in their relationships.
Legal Use of the Comprehensive Relationships With Industry Table ACCFHRS
The legal framework governing the usage of this table emphasizes transparency and ethical standards in participant-industry relationships. As per U.S. regulations, it helps identify potential conflicts of interest. The data compiled in the table must be handled in compliance with privacy and confidentiality laws to protect the sensitive commercial and financial information disclosed by participants.
Who Typically Uses the Comprehensive Relationships With Industry Table ACCFHRS
Primarily, the table is used by healthcare organizations, research institutions, and regulatory bodies to ensure transparency in guidelines and decision-making processes. Medical professionals and their support staff also rely on this documentation to uphold integrity and ethical standards in their professional engagements.


Required Documents
To accurately complete the table, gather several essential documents:
- Employment Contracts: For verifying employment status and industry roles.
- Consulting Agreements: To identify consulting relationships and remunerations.
- Financial Statements: Necessary for illustrating financial interests.
- Research Grants: Documents that highlight research funding and affiliations.
Why Should You Use the Comprehensive Relationships With Industry Table ACCFHRS
Utilization of this table is imperative for maintaining the integrity of Clinical practice guidelines. It helps identify potential biases that could arise from commercial influences. For the end user, including regulatory agencies and medical institutions, it provides a clear view of possible conflicts, ensuring that medical guidelines remain unbiased and patient-centered.