Definition & Meaning
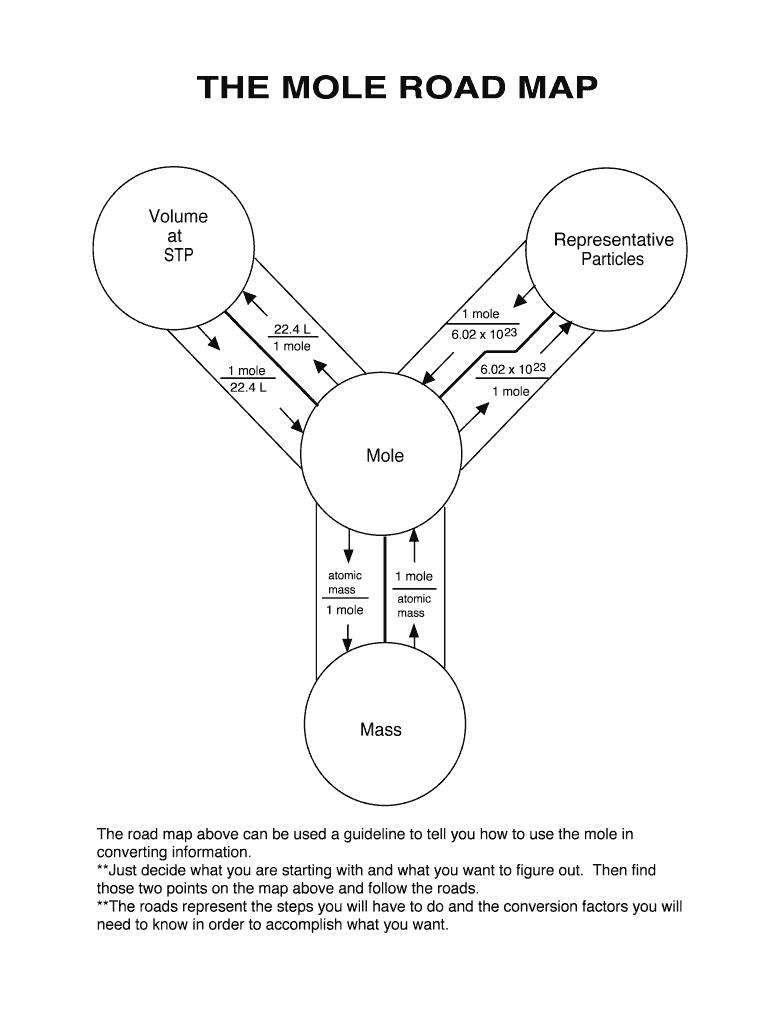
The Mole Road Map is a crucial educational tool in chemistry, designed to facilitate the conversion process relevant to moles. This map provides guidance on how to navigate conversions between various chemical quantities using foundational concepts such as Avogadro's number and the volume of one mole. By visually mapping out these relationships, The Mole Road Map simplifies the often complex processes involved in chemical equations and stoichiometry.
How to Use The Mole Road Map
The Mole Road Map supports students and professionals in calculating conversions between moles, particles, and volume. To begin, users must identify their starting point, such as the number of particles, and the target point, like the mole volume. Following the map's path, which includes steps like using Avogadro's number for conversion, ensures accurate and efficient calculations. This approach helps avoid errors in stoichiometric computations by offering a systematic process.
How to Obtain The Mole Road Map
Typically distributed in educational settings, The Mole Road Map can be accessed through textbooks, educational websites, or downloaded from academic resources. Instructors might provide printed copies or digital formats as part of chemistry courses. Availability varies based on the institution, but resources are generally accessible to help chemistry students and educators.
Steps to Complete the The Mole Road Map
- Identify Starting Quantity: Determine the initial chemical quantity, such as mass or volume.
- Locate Target Conversion: Establish the desired conversion outcome; for example, converting to particles.
- Select Conversion Factor: Choose the appropriate conversion factor; Avogadro's number for particles or 22.4 L for gases at standard temperature and pressure.
- Follow Conversion Path: Use the visual guide to apply the necessary conversion factors step by step.
- Verify Calculations: Check calculations against known chemical rules for accuracy.
Why Use the The Mole Road Map
Employing The Mole Road Map reduces the complexity of converting chemical quantities, ensuring accurate results. It is especially beneficial in academic settings for simplifying stoichiometric problems, helping students build a robust understanding of mole-related calculations. This tool is also indispensable for professionals needing precision in chemical preparations.
Key Elements of The Mole Road Map
- Avogadro's Number (6.02 x 10^23): Essential for converting between moles and particles.
- Molar Volume (22.4 L at STP): Used for converting moles to volume in gas calculations.
- Conversion Pathways: Visual guides connecting different chemical quantities through conversion factors.
- Reference Points: Clearly labeled starting and ending points for conversions, ensuring easy navigation of the map.
Important Terms Related to The Mole Road Map
- Stoichiometry: The calculation of reactants and products in chemical reactions.
- Molar Mass: The mass of one mole of a substance, critical for conversions involving mass.
- Standard Temperature and Pressure (STP): A reference point used in calculating gas volumes.
- Conversion Factor: A ratio used to convert one unit of measurement to another.
Examples of Using The Mole Road Map
A chemistry student working on a lab experiment might need to determine how many particles are present in a 1 mole sample of a substance. Using The Mole Road Map, they identify that 1 mole is equivalent to Avogadro’s number of particles, facilitating the conversion process. Another scenario might involve converting the volume of a gas to moles, where the map guides them using the 22.4 L molar volume at STP.
Legal Use of The Mole Road Map
Though an educational tool without direct legal implications, accuracy in using The Mole Road Map is vital for compliance in laboratory and industrial settings. Proper chemical conversions ensure safety, regulatory compliance, and integrity in research and commercial applications.