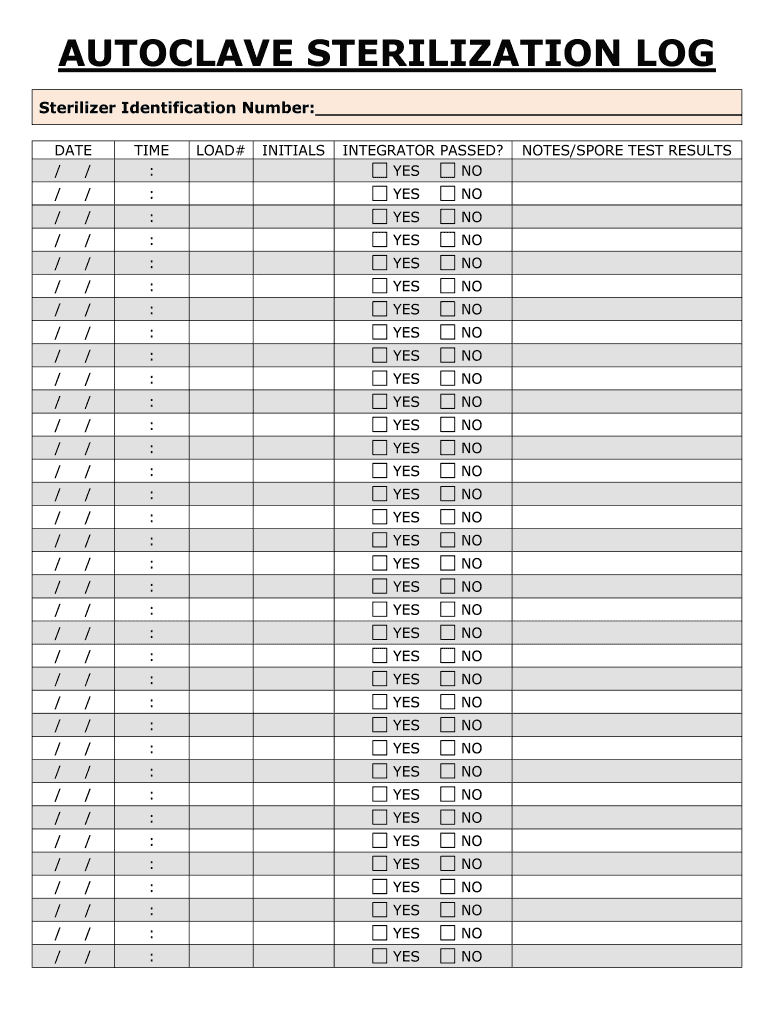
Definition and Meaning of the EBAP Autoclave Sterilization Log
The EBAP Autoclave Sterilization Log is a critical tool used in healthcare and laboratory settings to document the sterilization process of equipment and instruments. This log is essential for ensuring compliance with safety standards, maintaining the quality of sterilization, and keeping a meticulous record of sterilization cycles. Key information documented typically includes:
- Sterilizer identification number: This unique identifier helps track the specific autoclave used for sterilization.
- Date and time: The precise timing of each sterilization cycle is recorded to confirm that all procedures have been completed within designated times.
- Load number: Each batch or load being sterilized has a unique number associated with it, facilitating better tracking and management.
- Operator initials: The initials of the person operating the autoclave verify who performed the sterilization, creating accountability.
- Integrator results: This indicates whether the sterilization process was successful or if anomalies occurred during the cycle, confirmed by the integrator's performance.
The EBAP log not only serves as a compliance document but also as an audit tool that can help identify any issues with sterilization procedures and support quality control initiatives.
How to Use the EBAP Autoclave Sterilization Log
Utilizing the EBAP Autoclave Sterilization Log effectively involves following several important steps. These ensure that all necessary information is captured accurately, and the log fulfills its purpose as a verification document for sterilization processes.
-
Preparation: Before beginning sterilization, ensure the autoclave is in proper working condition. Gather all items that require sterilization and confirm they are compatible with the autoclave settings.
-
Filling Out the Log:
- Start by entering the sterilizer identification number at the top of the log.
- Record the date and time the sterilization cycle starts.
- Write the load number corresponding to this specific cycle.
- Ensure the operator’s initials are documented to establish accountability.
- After completing the sterilization cycle, check the integrator used and note whether it passed or failed.
- Add any relevant notes about the process or any events that occurred during the cycle.
-
Post-Sterilization Review: After the log is filled out, review all entries for accuracy. This may include verifying that the integrator results match the expected outputs based on the sterilization conditions.
-
Storage and Accessibility: Store the completed log in a secure yet accessible location for future reference. This can be critical in case of audits or when assessing sterilization effectiveness over time.
-
Regular Audits: Conduct periodic audits to ensure that logs are filled correctly and consistently. This helps identify any areas for improvement in sterilization techniques and log usage.
Proper use of the EBAP Autoclave Sterilization Log can enhance operational efficiency and reinforce compliance with safety regulations.
Key Elements of the EBAP Autoclave Sterilization Log
When dealing with the EBAP Autoclave Sterilization Log, several key elements must be accurately represented to ensure comprehensive tracking and accountability. Essential components include:
- Sterilization Cycle Information:
- Date and time of sterilization
- Load number to classify batches of items
- Sterilizer identification number for tracking purpose
- Operational Details:
- Initials of the operator to endorse accountability
- Comments or notes that highlight any particular circumstances during the process
- Quality Assurance:
- Integrator results, which indicate the effectiveness of the sterilization process
- Required spore test results to validate sterilization cycles according to standards
These elements contribute not only to operational transparency but also to the legal compliance required in many healthcare and research environments.
Important Terms Related to the EBAP Autoclave Sterilization Log
Understanding specific terminology related to the EBAP Autoclave Sterilization Log is essential for effective use and compliance. Familiarity with these terms can assist operators in better managing sterilization protocols:
- Integrator: An indicator used during sterilization that confirms the appropriate conditions were met (e.g., temperature and pressure).
- Cycle: The process during which an autoclave operates to sterilize instruments and supplies.
- Load: The collection of items placed within the autoclave during a single sterilization cycle.
- Spore Test: A method to validate the effectiveness of a sterilization cycle by using bacterial spores that are resistant to the sterilization process.
- Compliance: Adherence to institutional standards and governmental regulations regarding sterilization practices.
Clearly defining and understanding these terms ensures proper communication and enhances the effectiveness of sterilization programs within various healthcare settings.
Why You Should Use the EBAP Autoclave Sterilization Log
The EBAP Autoclave Sterilization Log serves multiple critical purposes that justify its use in any facility relying on sterilization processes. Key reasons include:
- Compliance Assurance: Maintaining an accurate log is often a regulatory requirement, ensuring that the facility adheres to health and safety regulations.
- Quality Control: The log aids in monitoring the effectiveness of sterilization processes over time, enabling facilities to ensure consistent quality in patient care and environmental safety.
- Risk Management: It helps identify trends or potential issues in sterilization efficacy, thus reducing risks associated with improper sterilization of instruments which could lead to infections.
- Accountability: Documenting who performed each sterilization cycle promotes responsibility among staff members and encourages adherence to best practices.
- Audit Preparedness: In many regulated environments, having meticulous records available facilitates smoother audits by external reviewers or regulatory bodies.
Using the log effectively is integral for maintaining operational standards and ensuring patient safety.
Steps to Complete the EBAP Autoclave Sterilization Log
Completing the EBAP Autoclave Sterilization Log properly involves a series of well-defined steps. These ensure clarity and accuracy in tracking sterilization processes:
-
Enter the Date and Time:
- Begin by documenting the correct date and time when the sterilization cycle is initiated.
-
Document the Load Number:
- Assign an appropriate load number for tracking purposes. Each cycle should have a unique identifier.
-
Input Sterilizer Identification Number:
- Record the specific autoclave's identification number that is utilized for the sterilization process.
-
Operator Identification:
- Include the initials of the operator responsible for the sterilization to promote accountability.
-
Record Integrator Results:
- After the sterilization process, document the results of the integrator. Indicate whether it passed or failed the cycle.
-
Additional Notes:
- Leave space for any relevant notes that may impact future processes, such as equipment malfunctions or observations during the cycle.
-
File for Future Reference:
- Once complete, ensure that the log is filed away securely but remains accessible for quick retrieval if needed for audits or reports.
Following these steps guarantees that the EBAP Autoclave Sterilization Log remains a reliable documentation tool in healthcare and laboratory settings.