



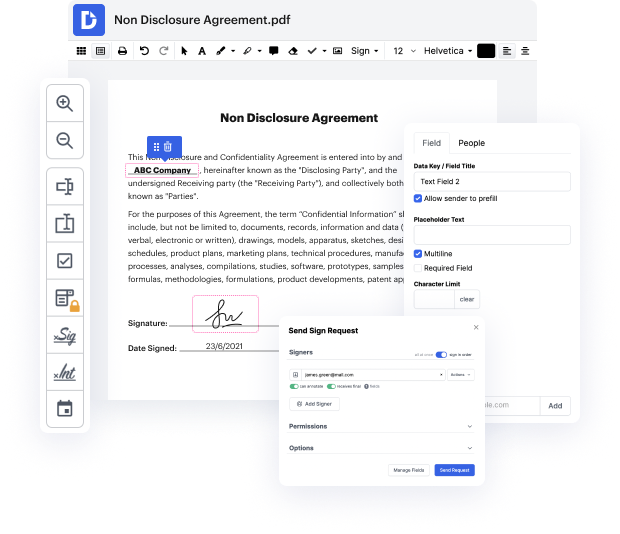
You know you are using the right document editor when such a simple task as Finish equation notice does not take more time than it should. Editing papers is now an integral part of a lot of working processes in numerous professional fields, which explains why convenience and simplicity are essential for editing instruments. If you find yourself studying tutorials or searching for tips on how to Finish equation notice, you may want to get a more easy-to-use solution to save your time on theoretical learning. And here is where DocHub shines. No training is needed. Just open the editor, which will guide you through its main functions and features.
A workflow becomes smoother with DocHub. Make use of this tool to complete the paperwork you need in short time and get your productivity to a higher level!
in this video were going to talk about how to write net ionic equations so lets consider the reaction between aqueous lead to nitrate and aqueous potassium chloride now the first thing that you need to do is you need to predict the products of this chemical reaction thats the first thing now what we have here is a double replacement reaction lead is going to pair up with chlorine and potassium is going to pair up with nitrate potassium has a positive one charge nitrate has a negative one charge if the magnitude of the charges are the same then you can combine those ions in a one-to-one ratio so one of the products will be kno3 now the other product we need to pair up lead and chloride lead has a positive two charge and we could tell based on how many nitrate ions are attached to it each nitrate ion has a minus one charge and theres two of them so to neutralize the two nitrate ions lead has to have a positive two charge chloride has a minus one charge so when you combine these two
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



