



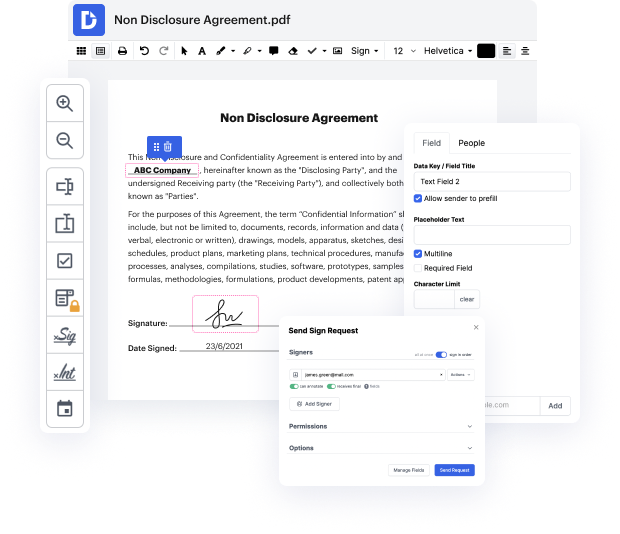
When you need to apply a minor tweak to the document, it must not take long to Finish equation accredetation. Such a simple activity does not have to require additional training or running through handbooks to learn it. Using the right document modifying instrument, you will not spend more time than is necessary for such a quick edit. Use DocHub to simplify your modifying process regardless if you are an experienced user or if it’s the first time making use of a web-based editor service. This tool will require minutes or so to learn how to Finish equation accredetation. The only thing needed to get more productive with editing is actually a DocHub profile.
A plain document editor like DocHub will help you optimize the amount of time you need to dedicate to document modifying irrespective of your previous experience with such instruments. Make an account now and improve your productivity immediately with DocHub!
For this example we will predict the products, balance the chemical equation, then using a solubility table well identify the solubility of each compound, then well find the complete ionic equation and finally well find the net ionic equation. Alright, lets predict our products. So were given our reactants here and the concept of the inner with the inner and outer with the outer, what I mean by that is the inner elements will combine to form our product and then so will the outer elements will combine to form our other product. So with the inner with the inner, well start off with lead and chlorine. You always want to put the positive charge, which is the metal and then the negative charge which is the nonmetal, together. So always put the metal first then the nonmetal. And then we know that lead is a 2+ charge and chlorine is a -1 charge. So what will happen is this two is going to come down and go with our chlorine because it has to be our subscript. so our first compound or o
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



