



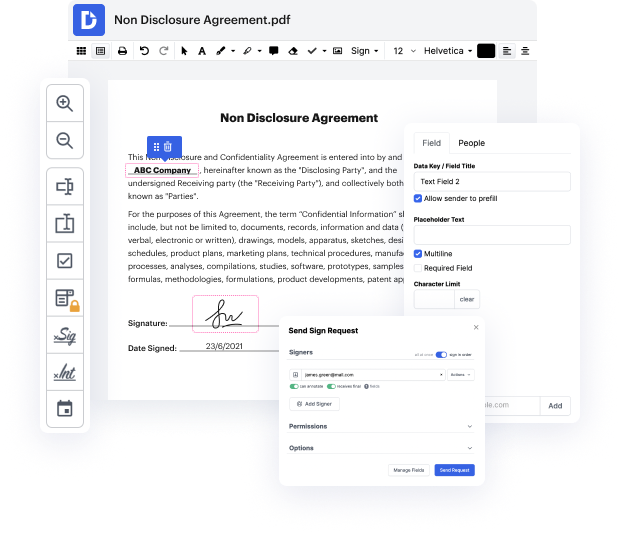
Are you having a hard time finding a reliable option to Conform Signature Article For Free? DocHub is set up to make this or any other process built around documents much easier. It's straightforward to navigate, use, and make changes to the document whenever you need it. You can access the essential features for dealing with document-based workflows, like certifying, adding text, etc., even with a free plan. Moreover, DocHub integrates with different Google Workspace apps as well as solutions, making file exporting and importing a breeze.
DocHub makes it easier to work on paperwork from wherever you’re. Additionally, you no longer need to have to print and scan documents back and forth in order to certify them or send them for signature. All the essential features are at your fingertips! Save time and hassle by executing paperwork in just a few clicks. Don’t hesitate another minute and give DocHub {a try today!
21 CFR part 11 is a set of rules that allows electronic records and signatures to be equivalent to paper records and handwritten signatures. This means you can replace paper documents and handwritten signatures with an electronic system and still comply with FDA regulations. The focus of this video is on the requirements for signing documents electronically, such as personal username and password, verification of the individual's identity, and notifying the FDA about the use of electronic signatures. Users must use two components - username and password - when signing records with electronic signatures.
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



