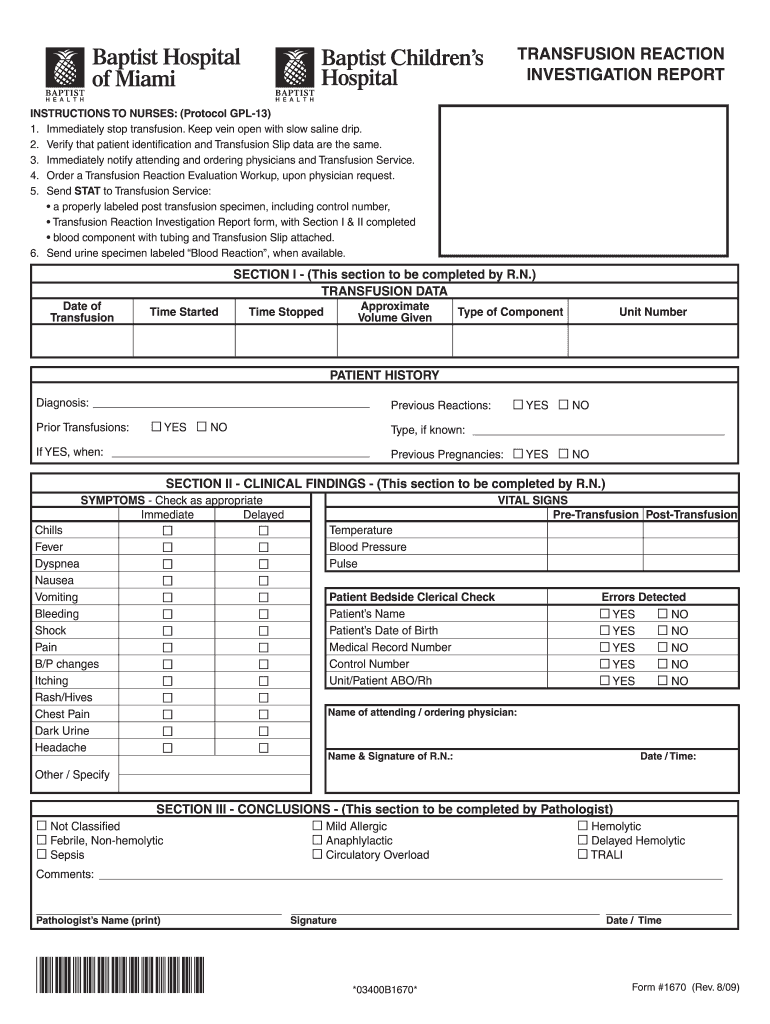
Definition and Importance of Transfusion Reaction GPL
Transfusion reaction GPL refers to the set protocols and forms used to document and investigate any adverse reactions that may occur during or after a blood transfusion. These reactions can range from mild allergic responses to severe complications such as hemolytic reactions, which can be life-threatening. Understanding and using the transfusion reaction GPL is critical for healthcare professionals to ensure patient safety, accurate documentation, and compliance with medical regulations.
The GPL provides a standardized approach to handling transfusion reactions by defining essential data elements to record, such as patient demographics, blood product details, clinical symptoms observed, and immediate actions taken. Implementing this form universally helps in tracking reactions, facilitating communication among medical staff, and providing necessary data for further evaluation and quality control in transfusion services.
- Documentation Importance: Accurate documentation in the transfusion reaction GPL serves as a legal record and assists in understanding the nature and frequency of transfusion reactions at healthcare facilities.
- Impact on Patient Care: Identifying and documenting adverse reactions can lead to prompt and appropriate medical responses, improving patient outcomes and potentially identifying issues with blood products.
Steps to Complete the Transfusion Reaction GPL
Completing the transfusion reaction GPL involves a systematic approach to ensure accurate and comprehensive documentation. Here are the detailed steps healthcare providers should follow:
-
Stop the Transfusion Immediately: As soon as a reaction is suspected, the transfusion should be halted to prevent further complications.
-
Verify Patient Information: Confirm the patient’s identity by checking their wristband and matching it with the blood product identification.
-
Notify Medical Personnel: Alert the physician and other relevant healthcare staff about the potential reaction to initiate further evaluation and treatment.
-
Collect Necessary Specimens: Obtain blood samples and any required laboratory specimens to identify the cause of the reaction. This may include a blood sample from the patient and the remaining blood bag if applicable.
-
Complete the Transfusion Reaction GPL Form: Document all observed symptoms, vital signs, and any interventions performed. Use standardized terms for clarity.
-
Report Findings: Submit the completed form to the appropriate department for review and further investigation.
-
Follow Up with the Patient: Monitor the patient closely and document any changes in their condition, ensuring thorough medical care throughout the process.
This systematic response to transfusion reactions not only safeguards patient health but also ensures compliance with hospital protocols and regulatory requirements.
Key Elements of the Transfusion Reaction GPL
Understanding the key elements involved in the transfusion reaction GPL allows healthcare professionals to use the form effectively and thoroughly. The following are essential components of the GPL:
- Patient Identification: Accurate details such as name, medical record number, and date of birth are crucial for clear documentation.
- Transfusion Details: Information on the blood product, such as type, lot number, expiration date, and transfusion start and end times.
- Symptoms Observed: Document specific clinical signs experienced by the patient, including fever, chills, rash, or any serious adverse reactions like hemolysis.
- Clinical Response: Report any immediate actions taken, such as administering medications, monitoring vital signs, and the response to those actions.
- Reporting and Review Process: Include a section for healthcare professionals to outline the steps taken in response to the reaction, including notifications made to the blood bank or risk management.
These elements form the backbone of the GPL, ensuring that the potential risks associated with transfusions are appropriately monitored and recorded.
Legal Use of the Transfusion Reaction GPL
The transfusion reaction GPL is not only a clinical tool but also serves legal purposes within the healthcare setting. Proper documentation can protect medical personnel and institutions from legal liability associated with transfusion reactions by providing a clear account of events and actions taken. Key factors include:
-
Compliance with Regulations: Many states and federal regulations demand accurate record-keeping of blood transfusions and reactions to ensure patient safety.
-
Legal Evidence: In case of adverse outcomes, a well-documented GPL can serve as evidence in legal cases, showing adherence to established protocols and helping to clarify the nature of the incident.
-
Reporting Requirements: Some jurisdictions may require that transfusion reactions be reported to health authorities, necessitating accurate and detailed documentation.
Healthcare providers must familiarize themselves with the legal implications of documentation and ensure that they complete the transfusion reaction GPL thoroughly to fulfill both clinical and legal obligations.
Examples of Using the Transfusion Reaction GPL
To illustrate the practical application of the transfusion reaction GPL, consider the following scenarios:
-
Mild Allergic Reaction: A patient develops hives and itching during a transfusion. After stopping the transfusion, the staff documents the reaction in the GPL, including the patient's vital signs and the immediate interventions, such as administering antihistamines.
-
Severe Hemolytic Reaction: A patient experiences acute back pain and fever during transfusion. The GPL would note the rapid response protocol initiated, including the collection of samples and immediate notification to the blood bank.
-
Post-Transfusion Fever: A patient develops fever several hours after completing a blood transfusion. Documentation in the GPL highlights the need for further investigation and monitoring, ensuring that follow-up actions are taken.
These examples underscore the importance of the transfusion reaction GPL in capturing a variety of potential reactions, thereby providing clarity and facilitating further medical response and inquiries.
With these detailed insights, healthcare professionals can navigate the complexities of transfusion reactions and ensure optimal patient safety through proper documentation and response protocols.