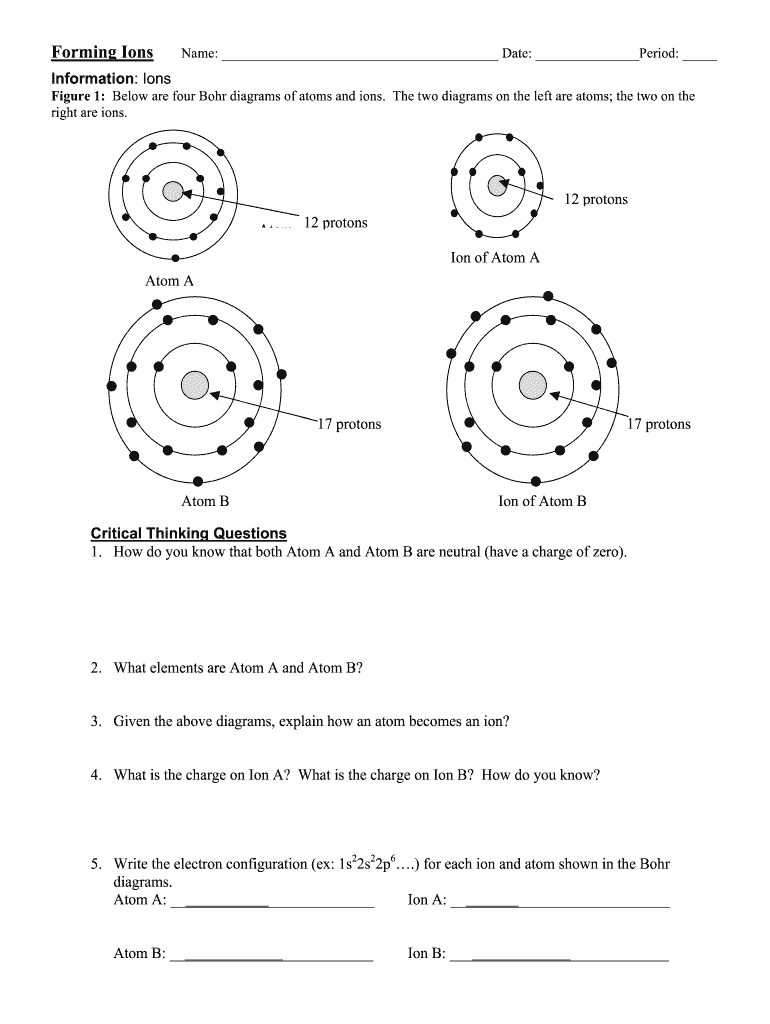
Definition and Meaning of Ion Formation
Ion formation refers to the process through which atoms gain or lose electrons to achieve a charged state, resulting in the creation of ions. Ions play a critical role in various chemical reactions and biological processes. The formation of ions typically occurs to reach a more stable electron configuration, often aligning with the structure of noble gases, which are naturally inert due to their complete valence electron shells.
Types of Ions
- Cations: Positively charged ions that form when an atom loses one or more electrons. For example, a sodium atom loses one electron to become a sodium cation (Na⁺).
- Anions: Negatively charged ions that arise when an atom gains one or more electrons. Chlorine, for instance, can gain an electron to become a chloride anion (Cl⁻).
Understanding these concepts helps students complete their ion formation worksheets effectively by identifying how atoms transition into ions and the implications that follow.
How to Use the Ion Formation Worksheet
The ion formation worksheet serves as an educational tool for students to practice and solidify their understanding of ion formation through structured exercises. Users typically follow these steps to utilize the worksheet effectively:
- Review the Basic Concepts: Before diving into the worksheet, students should familiarize themselves with terms such as cations, anions, and electron configuration.
- Complete the Exercises: The worksheet will usually contain various exercises, including drawing Bohr diagrams of neutral atoms and their corresponding ions, calculating the electrons gained or lost, and answering critical thinking questions.
- Analyze and Reflect: After completing the worksheet, students should review their answers, especially parts that involve critical thinking, to deepen their understanding of how ions are formed and their properties.
This structured approach aids in reinforcing learning objectives and ensuring mastery of the subject matter.
Steps to Complete the Ion Formation Worksheet
Completing an ion formation worksheet requires a systematic approach to ensure comprehensive understanding. Follow these steps:
- Read the Instructions Carefully: Understanding the worksheet's requirements will guide your efforts effectively.
- Identify Neutral Atoms: Begin by identifying the elements and their corresponding atomic numbers. This information is crucial for determining the number of electrons in neutral states.
- Draw Bohr Diagrams: Visual representation aids comprehension. Draw the isotopes of neutral atoms, showing the number of protons, neutrons, and electrons.
- Determine Ion Charges: Based on the exercise’s instructions, decide whether the ion is a cation or anion, and calculate the total charge resulting from the loss or gain of electrons.
- Fill in the Worksheet: Input your findings, ensuring accuracy in electron configurations and charge notations.
Each step ensures that students engage with the material critically, encouraging a deeper grasp of atomic structure and ion formation.
Examples of Using the Ion Formation Worksheet
Practical examples enhance understanding by illustrating how ions form in real-world scenarios. Some examples include:
- Sodium Ion Formation: When sodium (Na) loses one electron, its charge becomes +1, resulting in a sodium ion (Na⁺). This process is presented in the worksheet through a diagram and related exercises.
- Calcium Ion Formation: Calcium (Ca) can lose two electrons to form a Ca²⁺ ion. Worksheets typically involve drawing the Bohr diagram of calcium in both its neutral and ionic states.
- Chloride Ion Formation: Chlorine (Cl) gains one electron to form a Cl⁻ ion. Worksheets often include exercises where students calculate charge changes and draw corresponding diagrams.
These examples contextualize theoretical knowledge and allow students to apply their understanding to diverse atomic interactions.
Important Terms Related to Ion Formation
Familiarity with key terminology is vital for students working on the ion formation worksheet. It incorporates several critical terms, including:
- Electron Configuration: Describes the distribution of electrons in an atom's shells, crucial for understanding how atoms achieve stability.
- Valence Electrons: Electrons located in the outermost shell of an atom that are involved in chemical bonding and ionization.
- Stability: The concept that atoms tend to achieve electronic configurations similar to noble gases, thereby becoming less reactive.
- Ionization Energy: The energy required to remove an electron from an atom, relating directly to how easily an atom can become an ion.
These terms lay the groundwork for interpreting worksheet tasks and analyzing atomic behavior.
Who Typically Uses the Ion Formation Worksheet
The ion formation worksheet is primarily utilized by:
- High School Chemistry Students: These learners engage with the worksheet as part of their curriculum to reinforce core topics in chemistry.
- Educators: Teachers use the worksheet as a resource for both classroom instruction and home assignments, helping students understand complex concepts.
- Tutors: Individual tutors leverage the worksheet to provide targeted assistance to struggling students, addressing gaps in knowledge related to ionic compounds and reactions.
- Self-Learners: Individuals seeking to bolster their understanding of chemistry concepts often utilize worksheets for personal study.
Having a diverse audience, the ion formation worksheet serves as an essential educational resource across different learning contexts.


Legal Use of the Ion Formation Worksheet
The ion formation worksheet can be considered a widely accepted educational exercise and is legally used in academic settings. Schools and institutions who adopt these worksheets typically ensure compliance with educational guidelines.
Fair Use Considerations
- Copyrighted Content: If worksheets incorporate copyrighted material, users must be careful to follow guidelines around reproduction and distribution.
- Attribution: Should a worksheet credit specific sources or authors, users must adhere to those attribution requirements by appropriately acknowledging contributions.
Following these guidelines ensures that the ion formation worksheet is used within a legally acceptable framework, fostering a respectful learning environment.