Definition & Meaning
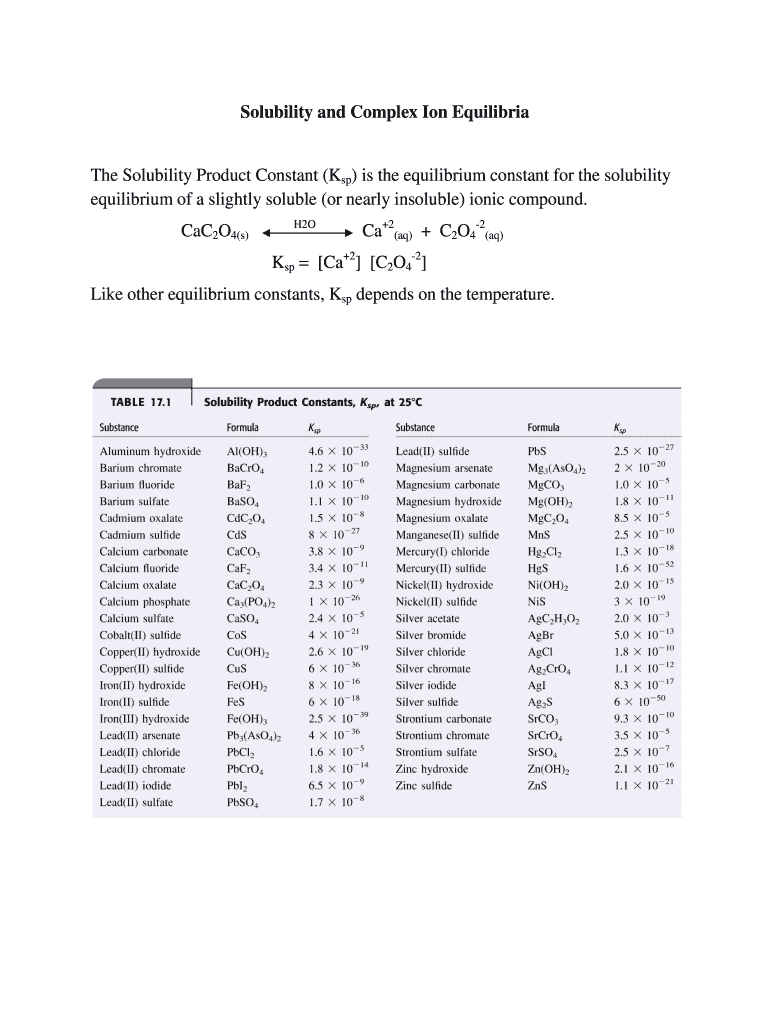
The equilibrium of a slightly soluble (or nearly insoluble) ionic compound refers to the state where the rate of dissolution of the compound equals the rate of precipitation in a saturated solution. This equilibrium is quantitatively described by the Solubility Product Constant (Ksp), which is a measure of the extent to which the compound can dissolve in water. The Ksp is specific to each compound and is dependent on temperature, offering insights into the solubility of substances under various conditions.
Important Terms Related to Equilibrium
Several key terms are essential when discussing the equilibrium of a slightly soluble ionic compound:
- Solubility Product Constant (Ksp): This is the equilibrium constant for the dissolution of a solid in a liquid, representing the maximum product of ionic concentrations that can coexist in a saturated solution.
- Precipitation Reaction: A process where dissolved ions combine to form an insoluble solid.
- Fractional Precipitation: A technique used to separate ions in solution by causing them to precipitate at different times.
- Complex Ion Formation: Occurs when simple ions in solution form complex, stability-ordering equilibria.
How to Use the Equilibrium of an Ionic Compound
Understanding the equilibrium of a slightly soluble ionic compound involves calculating the Ksp value to predict the solubility under given conditions. This calculation can help anticipate whether a precipitate will form when solutions are mixed. It is particularly useful in analytical chemistry for qualitative and quantitative analysis, enabling chemists to control reactions and processes effectively.
Steps to Complete the Equilibrium
To determine the equilibrium of a slightly soluble ionic compound, follow these steps:
- Identify the Compound: Determine the chemical formula of the ionic compound.
- Write the Dissolution Equation: Express the dissolution of the compound in its ionic form.
- Express Ksp: Write the Ksp expression using the concentrations of the resulting ions.
- Calculate Ksp: Use concentration data to calculate the Ksp or use given Ksp to find the maximum concentrations of ions.
- Evaluate Solubility: Compare with known solubility to verify precipitate formation.
Key Elements of Equilibrium
Several components make up the understanding of equilibrium in this context:
- Temperature Effects: Ksp varies with temperature, influencing the solubility of the compound.
- Ion Concentrations: The balance of products and reactants in a saturated solution influences solubility.
- Effect of pH: The solubility of some compounds increases under acidic or basic conditions due to additional reactions.
Examples of Using Equilibrium
Real-world applications include:
- Water Treatment: Managing precipitate formation to remove ions from water.
- Manufacturing: Controlling crystal formation in the production of materials.
- Pharmaceuticals: Ensuring the proper delivery and bioavailability of drugs by understanding compound solubility.
Who Typically Uses This Information
- Chemists and Researchers: For experimental planning and analysis.
- Environmental Scientists: In pollution control and water treatment processes.
- Pharmacists: To ensure product stability and efficacy.
Legal and Safety Considerations
Understanding the equilibrium process helps meet regulations regarding material safety data. Proper handling and disposal reduce environmental and health risks, ensuring compliance with legal standards related to chemical management.
State-Specific Rules for Equilibrium
Regulations can vary by state for industries reliant on these processes. Companies must stay informed about local legislation affecting solubility management, especially concerning environmental discharge and pollution control.
Digital vs. Paper Version
For precise, real-time analysis, digital tools offer dynamic calculation capabilities, integrating with laboratory software for streamlined processes. While traditional paper methods serve as reliable documentation, digital solutions facilitate faster adjustments and updates.