Definition and Meaning of the Protocol Signature Page Template
A protocol signature page template is a standardized document used primarily in clinical trials to ensure compliance and accountability among participants. This form is pivotal in solidifying the principal investigator's commitment to conducting a clinical study according to the established protocol, regulations, and Good Clinical Practice (GCP) guidelines. It encapsulates the agreement between the investigator and the trial sponsor, highlighting the importance of understanding and adhering to all investigative conditions and requirements.
Key Terms Explained
- Principal Investigator (PI): The researcher responsible for the conduct of the clinical trial.
- Protocol: A detailed plan outlining the study's methodology, objectives, and procedures.
- Good Clinical Practice (GCP): International standards established to ensure ethical and scientific quality in designing, conducting, and reporting trials.
How to Use the Protocol Signature Page Template
To effectively utilize the protocol signature page template, follow these steps:
- Review the Protocol: Ensure detailed understanding of the trial methodology, objectives, and any specific responsibilities outlined.
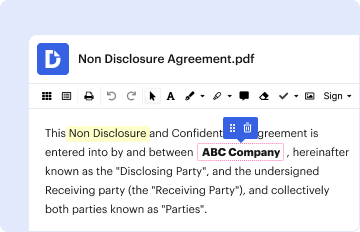
- Complete the Template: Fill in the required fields accurately, including the PI's name, trial ID, and date.
- Sign and Date: The primary investigator signs and dates the document to confirm their agreement and understanding.
- Submit to Sponsor: Send the completed form back to the trial sponsor or regulatory authority, as required.
Considerations for Use
- Ensure all information is accurate and matches the details in the trial protocol.
- Confirm whether any additional documentation needs to accompany the signature page.
Steps to Complete the Protocol Signature Page Template
Completing a protocol signature page template involves several methodical steps essential for maintaining compliance and ensuring clarity:
-
Verification of Details
- Double-check all identifying information such as trial identifiers, investigator's credentials, and study site location.
-
Understanding the Document
- Carefully read and comprehend all sections of the protocol provided by the sponsor.
-
Input Required Information
- Fill out necessary sections with precision, making sure all data corresponds with the protocol documentation.
-
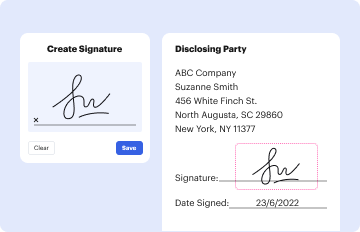
Sign and Date
- The PI must provide a legal signature and date the document, thereby confirming their understanding and agreement.
-
Store a Copy
- Retain a copy for personal records and for future reference in case of queries or audits.
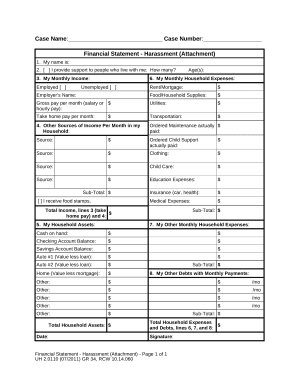
Key Elements of the Protocol Signature Page Template
The protocol signature page template contains several critical elements, each requiring careful attention:
- Investigator Information: Includes the name, contact details, and institutional affiliation of the PI.
- Trial Details: Provides the study title, protocol number, and key dates.
- Agreement Statements: Enumerates specific obligations and affirmations the PI acknowledges by signing.
- Signature Section: Space allocated for the PI's signature and date.
Additional Considerations
- Some templates may include sections for co-investigators or study coordinators.
- Certain trials may necessitate additional authorizations or waivers.
Who Typically Uses the Protocol Signature Page Template
Primarily, principal investigators in clinical trials use this template. However, the document may also be utilized by:
- Co-Investigators: Researchers assisting the PI in the trial.
- Study Coordinators: Professionals overseeing trial logistics and coordination.
- Regulatory Authorities: Bodies auditing or reviewing the clinical study's adherence to stipulated guidelines.

Applications in Different Scenarios
- Academic institutions involved in experimental drug evaluation.
- Medical centers conducting patient-centric clinical studies.
Legal Use of the Protocol Signature Page Template
The protocol signature page holds significant legal weight:
- Compliance Documentation: Establishes the PI's commitment to follow scientific and ethical guidelines.
- Regulatory Submission: Serves as vital evidence of compliance in submissions to regulatory bodies like the FDA.
Common Legal Considerations
- Ensure all signatories possess valid authority to commit to the terms.
- Be aware of specific laws or statutes that govern clinical trials in your region.
Software Compatibility
Most protocol signature page templates are structured to be compatible with a range of digital tools and platforms:
- Document Editing Software: Compatible with platforms like Microsoft Word and Google Docs.
- File Sharing Services: Easily integrated with cloud services such as Dropbox, Google Drive, and OneDrive.
- Digital Signature Platforms: Support electronic signing via DocHub, Adobe Sign, or other digital signature services.
Practical Use Cases
- Expedited document revision and collaboration via Google Workspace.
- Seamless transitions from document creation to signature collection using integrated platforms.
State-by-State Differences
In the context of the United States, state-specific regulations can influence the execution and submission of protocol signature pages:
- California and Massachusetts: States may have specific additional consent requirements.
- New York: Particular mandates related to data privacy in clinical trials may apply.
Key Variations
- Required additional submissions to state health departments.
- Differences in signature witnessing or notarization across states.
Each of these sections is designed to ensure users of the protocol signature page template have comprehensive guidelines and insights, thereby enhancing the integrity and efficiency of clinical trial processes.