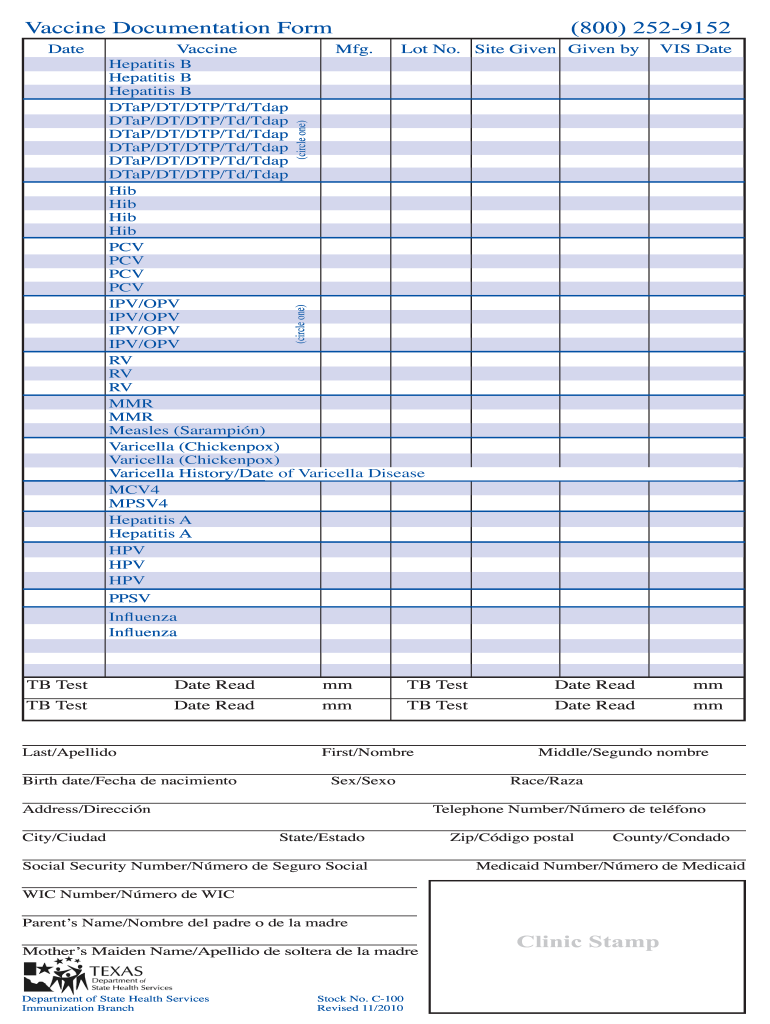
Understanding the Vaccine Documentation Form
The vaccine documentation form is a critical document for tracking immunizations administered to individuals. It typically captures essential details such as the type of vaccine received, the manufacturer, lot number, the site of administration, and the healthcare professional's credentials who administered the vaccine. This form serves as official acknowledgment of vaccination and is often required for school entry, travel, or employment in certain sectors.
Key Components of the Vaccine Documentation Form
-
Patient Information: This section includes personal details such as the recipient's full name, date of birth, and contact information. It ensures that the records accurately correspond to the individual receiving the vaccine.
-
Vaccine Details: This includes:
- Type of Vaccine: Specifies which vaccine was administered, such as MMR, TDap, COVID-19, etc.
- Manufacturer: Names the company that produced the vaccine.
- Lot Number: A unique identifier for quality assurance.
- Administration Site: Indicates where the vaccine was administered, typically an arm.
-
Healthcare Provider Information: Captures the signature and details of the administrator, including name, title, and contact information. This adds credibility and traceability to the vaccination process.
-
Consent and Acknowledgment: Recipients often must sign to acknowledge understanding of potential risks and benefits associated with vaccines. This section is crucial for legal and ethical documentation.
Purpose and Importance of Vaccine Documentation
Maintaining accurate vaccine records is paramount for several reasons:
-
School and Work Compliance: Many schools and employers require proof of vaccination before enrollment or employment. The documentation acts as assurance that individuals have received necessary vaccinations.
-
Public Health Tracking: Public health officials use vaccination records to monitor immunization rates in communities. This information helps to combat outbreaks of vaccine-preventable diseases and maintain herd immunity.
-
Personal Health Management: Individuals can reference their vaccination history during medical appointments or when required to provide proof for travel. Having a collected record helps in making informed health decisions.
How to Obtain and Use the Vaccine Documentation Form
The vaccine documentation form is typically provided by healthcare providers during an immunization visit. Additionally, many states offer downloadable versions that can be printed for personal use. To utilize the form effectively:
- Collect Information: Ensure that all relevant details are recorded accurately on the form.
- Review Consent Sections: Understand the implications of signing the consent acknowledgment, covering risks and benefits.
- Keep Copies: After receiving the vaccine, retain copies for your records. Having a digital version can be particularly useful for sharing easily when required.
Variations and State-Specific Rules
The specifics of the vaccine documentation form can vary by state, especially with regulations regarding the type of information collected. For instance:
- States may have unique immunization requirements for school attendance that necessitate specific vaccines documented on state-specific forms.
- Some states may offer a "Vaccine Information Statement" (VIS) along with the documentation, which details information about the vaccine's benefits and risks.
Additional Considerations for Vaccine Documentation
- Digital Versions: Many healthcare providers now offer electronic immunization records that can be accessed online. These digital versions may be deemed official by various institutions.
- ID Requirements: Some institutions may request identification, along with vaccine documentation, when confirming vaccination status.
The vaccine documentation form plays an essential role in managing individual health histories and ensuring compliance with public health regulations. Accurate completion and storage of this form can significantly impact one's educational, occupational, and travel-related opportunities.