



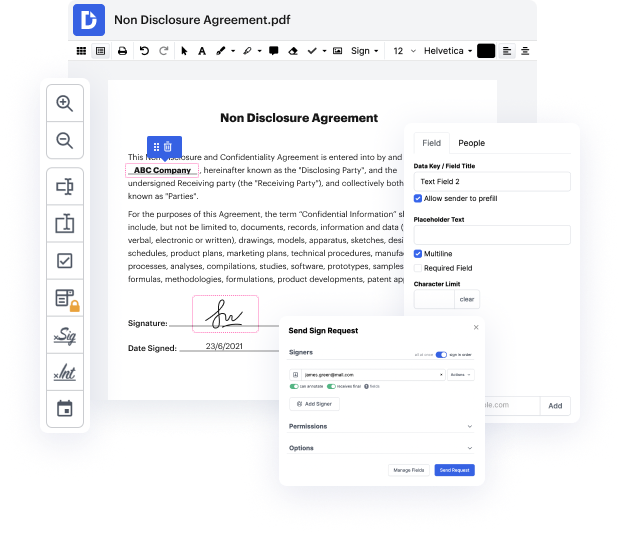
When your everyday work includes lots of document editing, you already know that every file format requires its own approach and often particular software. Handling a seemingly simple csv file can sometimes grind the whole process to a halt, especially if you are attempting to edit with insufficient software. To avoid this sort of problems, find an editor that will cover all your needs regardless of the file format and work in company in csv with zero roadblocks.
With DocHub, you will work with an editing multitool for virtually any occasion or file type. Reduce the time you used to invest in navigating your old software’s functionality and learn from our intuitive interface design while you do the job. DocHub is a sleek online editing platform that handles all of your file processing needs for virtually any file, including csv. Open it and go straight to productivity; no previous training or reading manuals is required to enjoy the benefits DocHub brings to document management processing. Start by taking a few minutes to create your account now.
See upgrades in your document processing immediately after you open your DocHub profile. Save your time on editing with our single solution that can help you become more productive with any file format with which you need to work.
hello friends welcome to pharma times napier silicon valley eros computer system validation csv in pharmaceutical industries computer system validation of the benefits entity computer system validation csv is a process used to endure that computer based systems will produce information or data that meet a set of defined requirements software pharmaceutical industrial operations foreign [Music] fda united states food and drug administration ich international conference of harmonization [Music] sorry ich recent international council of harmonization [Music] [Music] part level electronic records and electronic signatures validation of systems to ensure accuracy reliability consistent intended performance and the ability to [Music] 820 quality system regulation subpar c design control section 8 20.30 design validation shall include software validation and risk analysis where appropriate element design validation though software validation would include support g production and process con
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



