



COVID forever altered how companies see their internal protocols and procedures. It impacted organizations of all sizes and industries, posing new challenges for staying connected. The pandemic indicated that all companies should integrate digital instruments into day-to-day routines. They became essential for far more than hybrid working models.
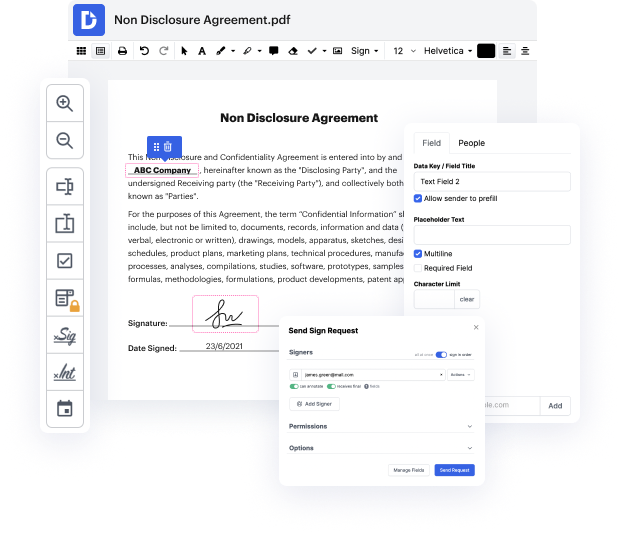
Platforms like DocHub enable you to increase your document management and approval operations. DocHub is the go-to instrument for end-to-end online editing and signatures. It helps reduce your day-to-day contract and agreement generation and approval tasks. Obtain access to Word Software for Medical Device Manufacturers | Medical Device Manufacturers Document Management Solution advanced editing features that cover all of your management requires. Work with any document type and formatting, produce fillable fields, and successfully collect signatures from your colleagues and customers. No past training or experience is required.
With Word Software for Medical Device Manufacturers | Medical Device Manufacturers Document Management Solution, you can maximize the quality of your files, increase the approval process, and safely store complete files. Obtain a free DocHub account right now and upgrade your plan when you want.
In this short course on design control for medical devices, Peter Sibelius, with 20 years of experience in medical device product development, shares key concepts such as intended use, user needs, design input, verification, validation, and transfer. This course is a starting point for those in product development, aiming to provide a basic understanding of design control's importance and potential benefits for career advancement. The full online course offers in-depth knowledge and skills for those interested in furthering their expertise in this field.
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



