



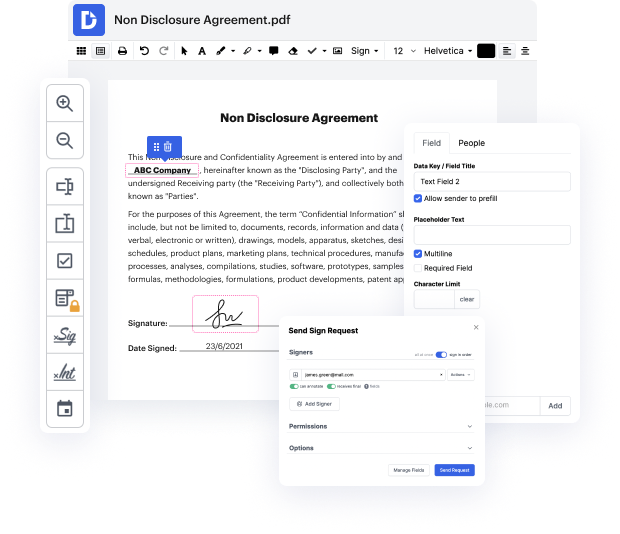
Are you searching for an editor that enables you to make that last-moment edit and Type Equation Notification For Free? Then you're in the right place! With DocHub, you can easily make any required changes to your document, no matter its file format. Your output files will look more professional and structured-no need to download any software taking up a lot of space. You can use our editor at the comfort of your browser.
When using our editor, stay reassured that your sensitive information is protected and shielded from prying eyes. We comply with significant data protection and eCommerce regulations to ensure your experience is safe and enjoyable every time! If you need help editing your document, our professional support team is always ready to answer all your questions. You can also benefit from our comprehensive knowledge center for self-assistance.
Try our editor today and Type Equation Notification For Free with ease!
The video discusses how to write net ionic equations by considering the reaction between aqueous lead nitrate and aqueous potassium chloride. To predict the products, it is important to understand that lead pairs up with chloride and potassium pairs up with nitrate based on their charges. Potassium nitrate (KNO3) and lead (II) chloride (PbCl2) are the products of this chemical reaction. Lead has a positive two charge, while chloride has a negative one charge, resulting in the formation of PbCl2 to neutralize the two nitrate ions attached to lead.
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



