



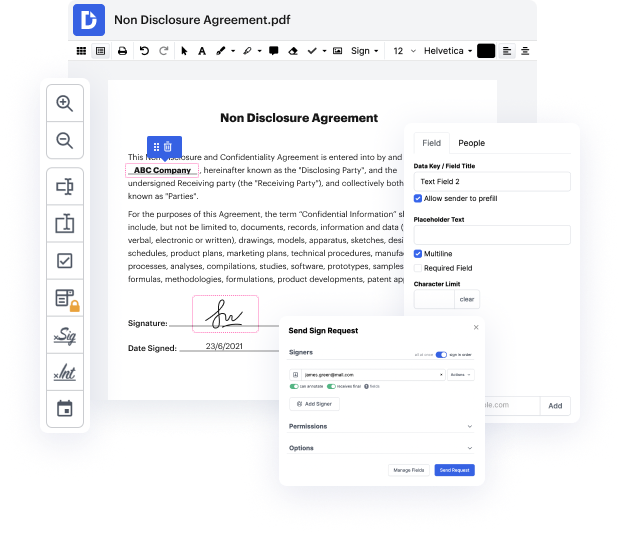
DocHub is a powerful online tool designed for seamless document editing, signing, and distribution. With its robust features, users can efficiently manage their documents directly from their ChromeBook. Not only does our platform integrate deeply with Google Workspace, but it also provides an easy way to track account activity using the Audit Trail feature, ensuring complete transparency and accountability in your document workflows.
Start using DocHub today to enhance your document management experience and track account activity effortlessly!
hi friends welcome to our channel 21 cfr part 11. please subscribe our youtube channel and hit the bell button to get more such updates in this video we are going to see about the electronic audit trail review [Music] the usfda21cfr part 11 and eugmp nx11 clause 9 requests that the consideration should be given based on a risk assessment to building into the system the creation of a record of all gmp relevant changes and deletions in short a system generated audit rail for change or deletion of gmp relevant data the reason should be documented audit trail need to be available and convertible to a generally intelligible form and regularly reviewed now as you have already seen 21 cfr part 11 and eu gmp nx11 clause 9 request now what if the request is not followed youre going to get a warning now the warning is as follows heres the example for you your firms review of laboratory data does not include a review of an audit trail or revision history to determine if unapproved changes have
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



