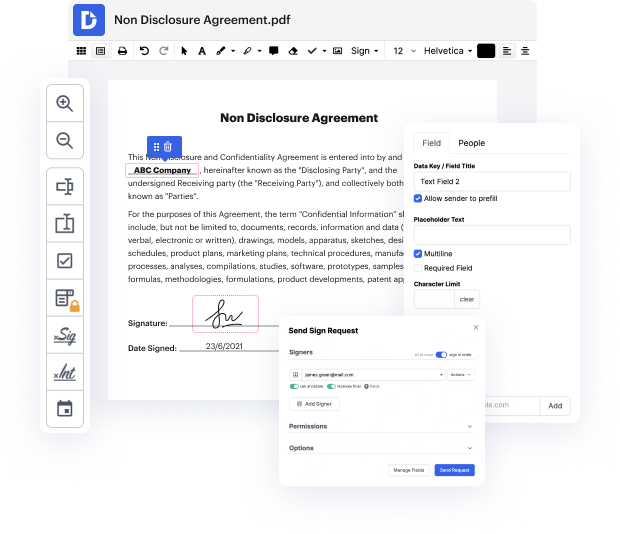
Handling papers like Manufacturing Contract may seem challenging, especially if you are working with this type for the first time. Sometimes a little edit might create a major headache when you don’t know how to work with the formatting and avoid making a chaos out of the process. When tasked to tack trace in Manufacturing Contract, you could always make use of an image modifying software. Other people might choose a conventional text editor but get stuck when asked to re-format. With DocHub, though, handling a Manufacturing Contract is not harder than modifying a document in any other format.
Try DocHub for fast and efficient papers editing, regardless of the file format you have on your hands or the type of document you need to fix. This software solution is online, reachable from any browser with a stable internet connection. Edit your Manufacturing Contract right when you open it. We’ve developed the interface so that even users with no prior experience can readily do everything they require. Simplify your paperwork editing with a single streamlined solution for just about any document type.
Dealing with different kinds of documents must not feel like rocket science. To optimize your papers editing time, you need a swift platform like DocHub. Manage more with all our tools at your fingertips.
The company is adapting to increasing regulatory demands and costs by offering value-added assembly solutions, including drug handling, cold chain management, serialization, and sterilization. These capabilities streamline supply chain logistics, improve operating efficiencies, and support regulatory guidelines. Customers are now required to comply with FDA's UDI requirement, which the company supports by ensuring data integrity for medical devices to enable track and trace.