People who work daily with different documents know very well how much efficiency depends on how convenient it is to access editing tools. When you Online Therapy Consent documents must be saved in a different format or incorporate complicated elements, it may be difficult to deal with them utilizing classical text editors. A simple error in formatting may ruin the time you dedicated to tack city in Online Therapy Consent, and such a basic job should not feel hard.
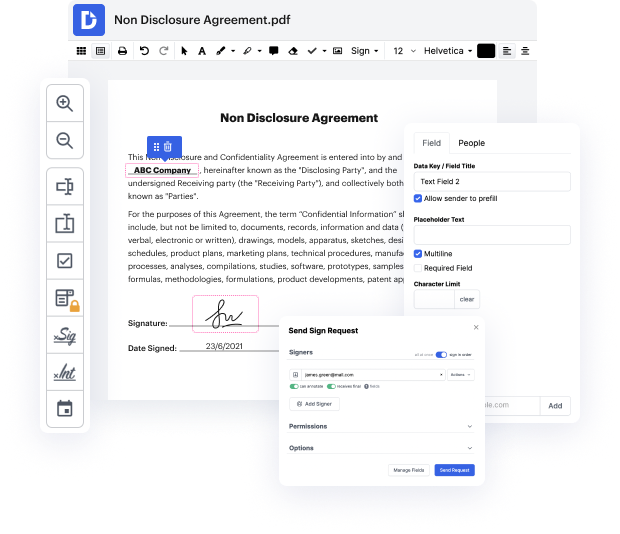
When you find a multitool like DocHub, such concerns will in no way appear in your work. This powerful web-based editing solution will help you easily handle paperwork saved in Online Therapy Consent. You can easily create, edit, share and convert your documents wherever you are. All you need to use our interface is a stable internet connection and a DocHub profile. You can create an account within minutes. Here is how simple the process can be.
Using a well-developed modifying solution, you will spend minimal time finding out how it works. Start being productive as soon as you open our editor with a DocHub profile. We will make sure your go-to editing tools are always available whenever you need them.
Before participating in the study, it is crucial to understand what it entails. Researchers will provide an informed consent document detailing the study's duration, visits, medications, procedures, outcomes, benefits, risks, and more. If needed, a translator will be available. After reviewing the document and discussing with staff and family, you can decide whether to participate. Signing the informed consent statement signifies your voluntary agreement. You can choose to leave the study at any time, even after signing the document.