



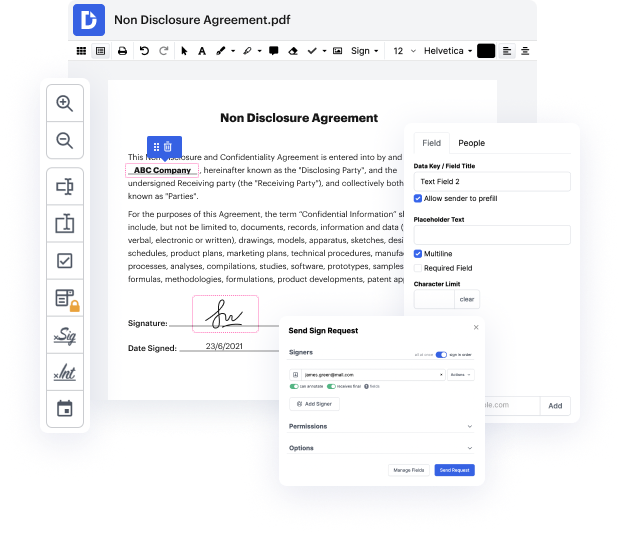
You know you are using the right document editor when such a basic task as Structure signature title does not take more time than it should. Modifying documents is now a part of many working processes in various professional fields, which explains why accessibility and straightforwardness are essential for editing resources. If you find yourself researching guides or searching for tips on how to Structure signature title, you may want to find a more intuitive solution to save time on theoretical learning. And this is where DocHub shines. No training is required. Simply open the editor, which will guide you through its main functions and features.
A workflow gets smoother with DocHub. Take advantage of this instrument to complete the files you need in short time and take your efficiency to another level!
21 CFR part 11 is a set of rules that specifies what is required for electronic records and signatures in order to be equivalent with paper records and handwritten signatures this means you can replace your paper based documentation and handwritten signatures with an electronic system and comply with FDA regulations first of all the requirements for signing documents electronically which is the focus of this video secondly the requirements for electronic record-keeping such as system validation and audit trail which well touch upon in another video these are the primary requirements in order to use electronic signature the user name and password are personal and cannot be used by anybody else the company must verify the identity of the individual that would be using the electronic signature the company must notify FDA about using electronic signatures when users use electronic signatures they must use two components username and password when signing records with electronic signature
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



