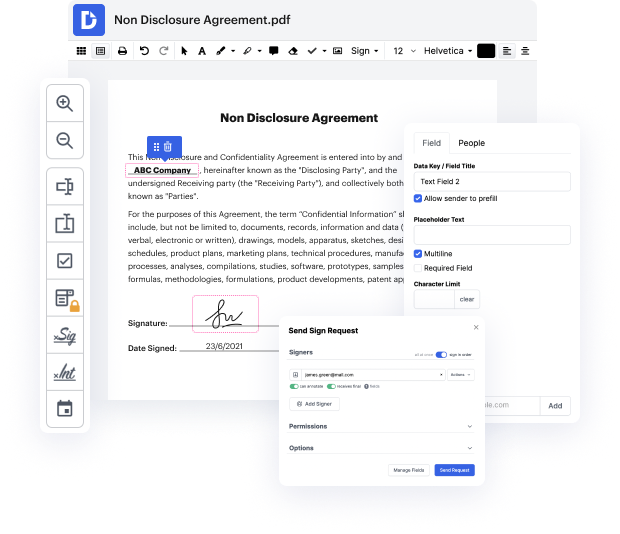
Document generation and approval are central components of your day-to-day workflows. These procedures are frequently repetitive and time-consuming, which effects your teams and departments. In particular, Quality Incident Record creation, storing, and location are significant to ensure your company’s productivity. A comprehensive online platform can resolve several vital concerns related to your teams' productivity and document management: it eliminates tiresome tasks, simplifies the process of locating documents and gathering signatures, and leads to more precise reporting and analytics. That is when you may need a strong and multi-functional platform like DocHub to deal with these tasks rapidly and foolproof.
DocHub allows you to make simpler even your most complicated process using its powerful features and functionalities. An excellent PDF editor and eSignature enhance your day-to-day document administration and make it the matter of several clicks. With DocHub, you will not need to look for additional third-party solutions to complete your document generation and approval cycle. A user-friendly interface lets you begin working with Quality Incident Record immediately.
DocHub is more than simply an online PDF editor and eSignature software. It is a platform that can help you easily simplify your document workflows and integrate them with well-known cloud storage solutions like Google Drive or Dropbox. Try out modifying Quality Incident Record immediately and discover DocHub's considerable list of features and functionalities.
Begin your free DocHub trial right now, with no concealed fees and zero commitment. Uncover all features and possibilities of seamless document administration done right. Complete Quality Incident Record, gather signatures, and increase your workflows in your smartphone app or desktop version without breaking a sweat. Increase all of your day-to-day tasks using the best solution accessible on the market.
so again um as uh i was mentioning um chris anderberg works at sccr as quality and compliance manager um she has over 30 years of research experience working for academic research organizations and industry her knowledge extends to clinical trial management for globe global studies gcp compliance audits regulatory agency inspections clinical event education quality management of clinical trials and policy and procedure development so thank you so much chris for being here with us and i appreciate your time and i hand it over to you you you are muted chris you are muted let me see if i can unmute you there that should be good perfect all right sorry about the technical difficulties ive had these twice this week um thank you very much susan for the very nice introduction im very excited to be here and i wanted to spend our time together talking about quality incident reporting and youll see as we go through were going to be talking about kappas and im hoping that by going through t