Unusual file formats in your daily papers management and editing processes can create instant confusion over how to modify them. You may need more than pre-installed computer software for effective and fast document editing. If you need to set formula in WRI or make any other simple alternation in your document, choose a document editor that has the features for you to work with ease. To handle all of the formats, such as WRI, choosing an editor that works well with all kinds of files will be your best choice.
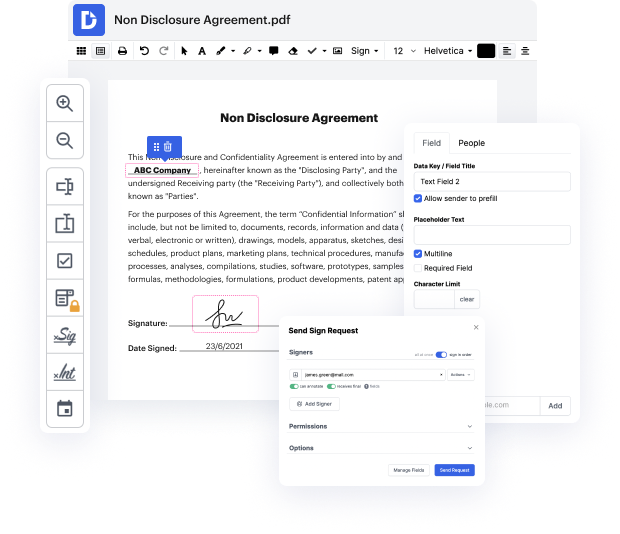
Try DocHub for effective document management, regardless of your document’s format. It has potent online editing instruments that streamline your papers management operations. You can easily create, edit, annotate, and share any document, as all you need to access these features is an internet connection and an functioning DocHub account. A single document tool is everything required. Don’t waste time jumping between different programs for different files.
Enjoy the efficiency of working with an instrument created specifically to streamline papers processing. See how easy it really is to modify any document, even when it is the first time you have dealt with its format. Register an account now and enhance your whole working process.
in this lesson were going to talk about how to write the chemical formulas of ionic compounds now before you do so you need to know the charges of certain ions so lets focus on the elements in group one like lithium sodium potassium these elements they have one valence electron and so they tend to form plus one charges or cations with positive one charges now in the second column you have the group two elements the alkaline earth metals and these include elements such as calcium magnesium and so forth these elements they form ions with a two plus charge now in the middle you have transition elements which can have variable charges so were not going to focus on those too much but moving on to group 13 also known as group 3a you have elements such as aluminum im going to write it over here and this element has a 3 plus charge and then you have elements like carbon silicon germanium which are found in group 4a and for ionic compounds its rare that youll see those elements in group