



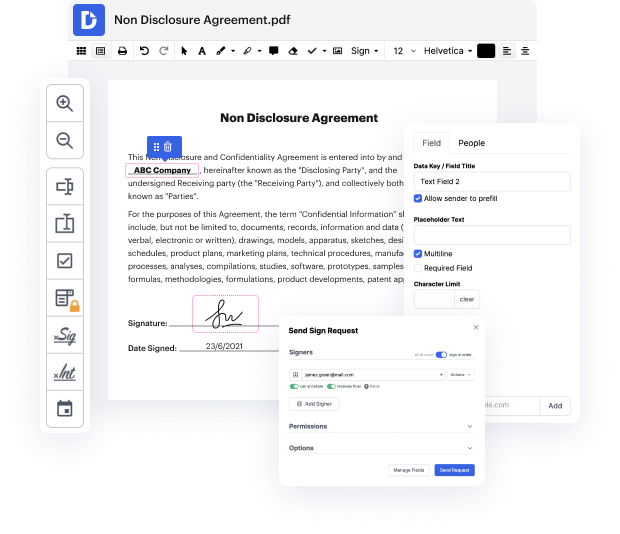
If you want to apply a small tweak to the document, it should not take long to Report form. This kind of basic activity does not have to demand additional education or running through manuals to understand it. Using the proper document modifying resource, you will not take more time than is needed for such a swift edit. Use DocHub to streamline your modifying process regardless if you are an experienced user or if it’s your first time using a web-based editor service. This tool will require minutes or so to figure out how to Report form. The sole thing needed to get more productive with editing is actually a DocHub account.
A plain document editor like DocHub can help you optimize the time you need to spend on document modifying no matter your prior experience with this kind of tools. Make an account now and improve your efficiency instantly with DocHub!
In clinical studies, essential documents include the study protocol, investigators brochure, and case report form. The trend is shifting towards using electronic case report forms (eCRF) for data collection to avoid illegible handwriting and reduce data queries. Remote data entry or electronic data capture is becoming more common in the research industry, leading to more efficient data management and less room for error in data recording.
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



