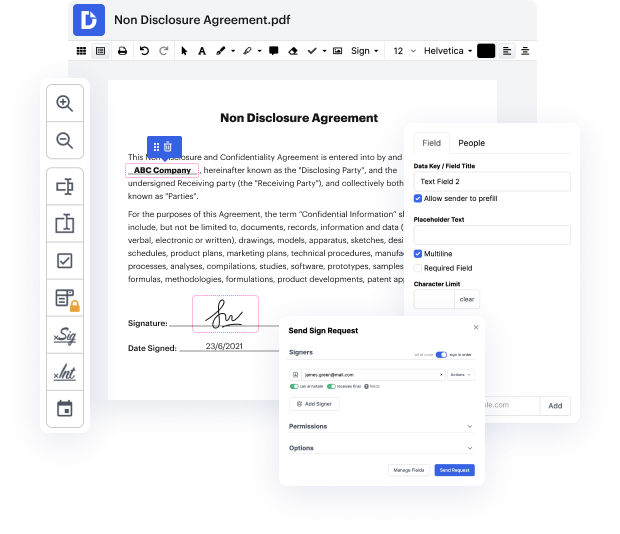
Time is an important resource that every enterprise treasures and attempts to transform into a gain. When picking document management software program, be aware of a clutterless and user-friendly interface that empowers customers. DocHub offers cutting-edge features to maximize your file managing and transforms your PDF editing into a matter of a single click. Replace Sticky Notes to the Clinical Trial Agreement with DocHub in order to save a lot of time as well as increase your efficiency.
Make PDF editing an simple and intuitive operation that will save you a lot of valuable time. Effortlessly change your files and send out them for signing without having looking at third-party alternatives. Give attention to pertinent tasks and increase your file managing with DocHub right now.
[Music] this is a demonstration of how to use the practical guidance life sciences practice area to prepare agreements related to the many facets of clinical research for drugs and medical devices one of the fastest and easiest ways to find tools for drafting clinical research agreements is through the clinical trials task on the practical guidance life sciences practice area page here you will find content focused on clinical trials you can get information about fundamentals including contract research organizations irbs and other matters for example this practice note provides drafting tips for contract research organization agreements it also includes a link to cro and research services agreements precedents that will help you to benchmark your agreements with those used by public companies through transaction search by intelligize whether you are tasked with drafting a clinical research support agreement a master clinical trial agreement a material transfer agreement or a sponsored