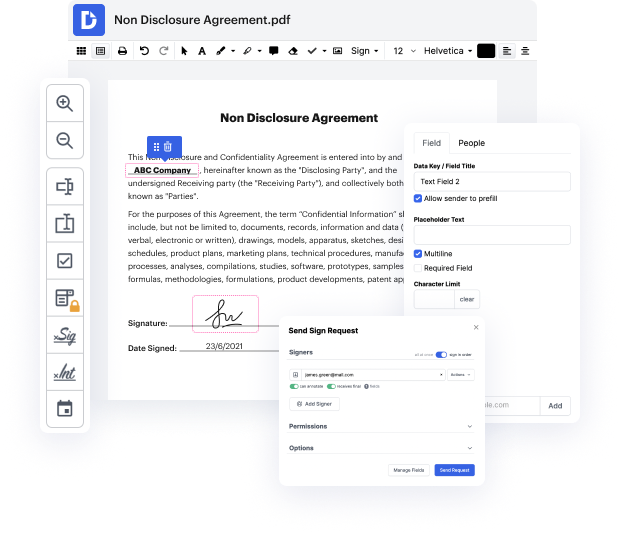
Time is a crucial resource that each organization treasures and attempts to convert in a reward. When choosing document management software, focus on a clutterless and user-friendly interface that empowers users. DocHub provides cutting-edge tools to maximize your file administration and transforms your PDF file editing into a matter of a single click. Replace Required Fields into the Product Defect Notice with DocHub in order to save a ton of time as well as improve your efficiency.
Make PDF file editing an simple and intuitive operation that saves you plenty of valuable time. Quickly adjust your documents and give them for signing without the need of adopting third-party options. Concentrate on pertinent duties and enhance your file administration with DocHub today.
the Health Sciences authority or HSE as a post market surveillance program to monitor the quality safety and efficacy of their building products in Singapore one component of this program is the management of product defects as the product registrant manufacturer importer or supply of therapeutic product you are required to do the following to report product defects related to your therapeutic product to HSA or to notify us if you are recalling any product before initiating the recall process this video aims to provide you with information regarding the requirements and the processes for further defect reporting and product recalls further defects can arise during the manufacturing the storage and the handling of therapeutic products it is important that you report these defects arising from during these processes especially if they can affect a product safety quality and efficacy this can help identify issues that may affect the users and ensure measures are taken to protect public he