Document generation is a essential aspect of successful firm communication and management. You need an affordable and efficient solution regardless of your papers preparation point. protocol preparation may be one of those operations which require extra care and focus. Simply explained, there are better possibilities than manually producing documents for your small or medium enterprise. Among the best ways to make sure good quality and usefulness of your contracts and agreements is to set up a multi purpose solution like DocHub.
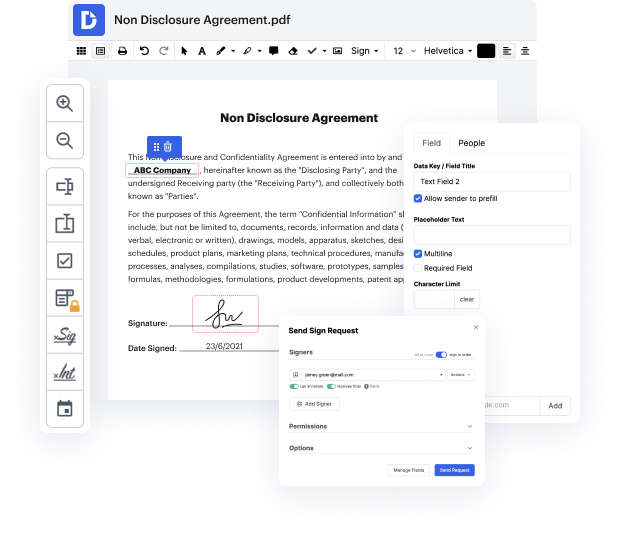
Editing flexibility is considered the most considerable advantage of DocHub. Use robust multi-use tools to add and take away, or change any aspect of protocol. Leave feedback, highlight important information, put in writing in protocol, and transform document managing into an simple and user-friendly procedure. Access your documents at any moment and apply new modifications whenever you need to, which could substantially lower your time making the same document completely from scratch.
Produce reusable Templates to make simpler your day-to-day routines and steer clear of copy-pasting the same information continuously. Modify, add, and modify them at any moment to ensure you are on the same page with your partners and clients. DocHub helps you steer clear of errors in frequently-used documents and offers you the very best quality forms. Make certain you always keep things professional and stay on brand with your most used documents.
Enjoy loss-free protocol editing and secure document sharing and storage with DocHub. Don’t lose any files or find yourself confused or wrong-footed when discussing agreements and contracts. DocHub enables specialists everywhere to adopt digital transformation as an element of their company’s change management.
hi and welcome to another lecture this is sort of a shorter one were gonna talk about the study protocol youve heard me mention this before but what exactly is a study protocol what do you use it for why do you have it so what exactly is a study protocol you ask thats an excellent question its a plan written by the sponsor which describes how study will be conducted and eventually how the data will be analyzed its the blueprint its its the recipe if you will for how youre gonna run a clinical research study it is designed around a single research question now let me sort of expand on this a little bit there will be a primary objective of every study sometimes sponsors will put in secondary interests so they might have one primary research question and if base ones study right itd be what are the adverse events but its a tolerability and what is the maximum tolerated dose they might have some other little questions in there like does this study drug improve improve pain for e