



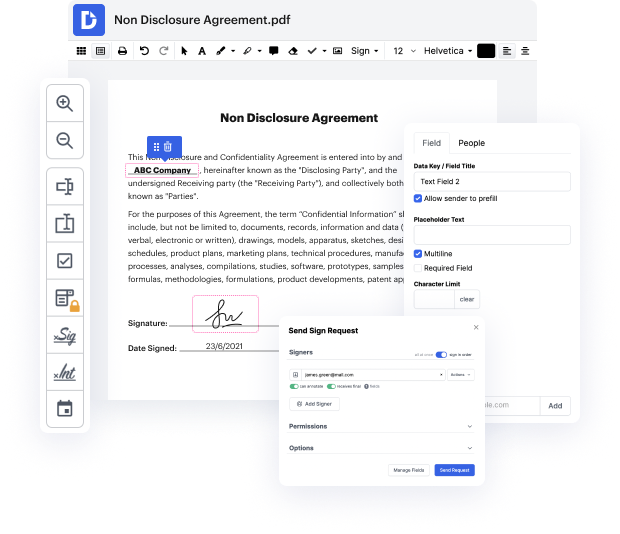
You know you are using the right file editor when such a basic task as Publish formula notification does not take more time than it should. Editing files is now an integral part of many working operations in different professional areas, which is why accessibility and efficiency are essential for editing instruments. If you find yourself studying manuals or searching for tips about how to Publish formula notification, you might want to find a more intuitive solution to save time on theoretical learning. And here is where DocHub shines. No training is required. Simply open the editor, which will guide you through its main functions and features.
A workflow becomes smoother with DocHub. Use this tool to complete the files you need in short time and take your efficiency one stage further!
good afternoon and welcome to a webinar on regulatory packages of new infant formula submissions hosted by EAS Consulting Group and presented by EAS independent advisor for food color additive safety dr. Robert Martin independent consultants dr. Robert Burns and dr. Timothy Mork and Senior Director for food consulting services Alan sailer EAS specializes in FDA regulatory matters with a prime focus of assisting domestic and foreign food pharmaceutical dietary supplement medical device tobacco and cosmetics firms comply with applicable laws and regulations EAS is staffed with former FDA compliance and inspection officials and industry executives and is assisted by an extensive network of consultants with many years of FDA and industry experience todays presenters are dr. Robert Martin who is a former deputy director for FDAs division of biotechnology and graphs notice review dr. Robert or Robbie Burns who was a former global nutrition and scientific affairs director for Cadbury Sch
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



