



Keeping up with the fast-transforming work environment that has emerged since COVID remains to be an essential aspect for so many sectors. Many organizations look for an accessible and easy-to-adopt platform that will be accessible for them 24/7. The key interest lies in addressing all file generation and approval processes with minimal effort and time. DocHub offers powerful features and storage integrations that can make positive changes to day-to-day file processes forever. You just need a free DocHub user profile to access PDF Application for Life Sciences | Life Sciences Document Management Solution.
DocHub is an end-to-end editing and annotation app that covers all of your document generation demands. Make documents completely from scratch, modify them, leave feedback, and quickly collect signatures from teammates and clients. Forget about losing your documents or fearing about security - DocHub has industry-leading security standards that safeguard your data.
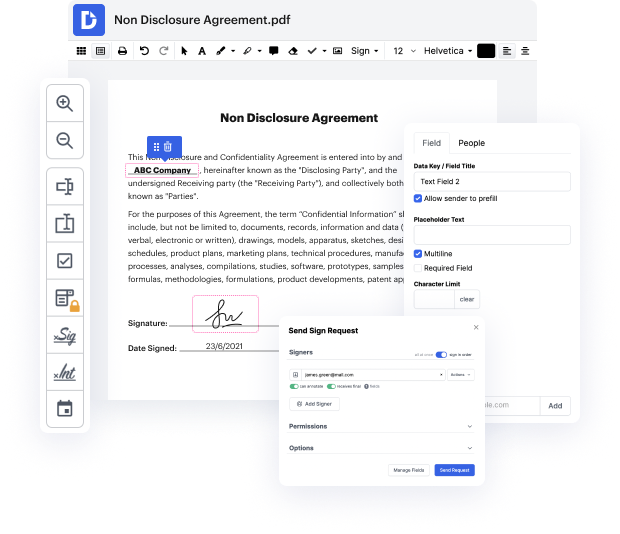
Focus on agreements and contracts anywhere, anytime. Get the most from PDF Application for Life Sciences | Life Sciences Document Management Solution and boost your day-to-day file management, from file creation to approval and storage.
Large, complex distributed organizations, especially in Life Sciences, face challenges with managing numerous documents spread across file shares, SharePoint sites, and even in print format. Without a clear understanding of the documents, their versions, ownership, and access, these organizations struggle to maintain control. Defined document management processes are crucial for managing policies, procedures, standards, and other operational documents in a life sciences company. Knowing document ownership, versions, and access is essential for success, as well as for compliance, risk management, and auditing purposes. An audit trail tracking document access is also necessary for transparency and control.
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



