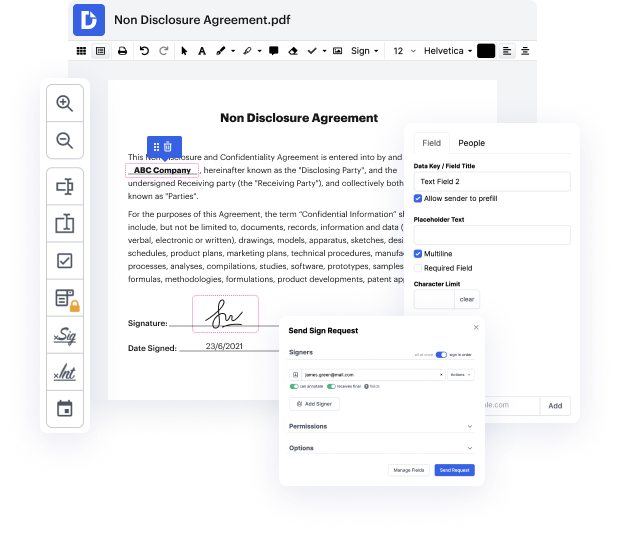
Of course, there’s no ideal software, but you can always get the one that perfectly combines powerful functionality, intuitiveness, and reasonable cost. When it comes to online document management, DocHub offers such a solution! Suppose you need to Modify header in Professional Medical Consent and manage paperwork efficiently and quickly. In that case, this is the suitable editor for you - complete your document-related tasks anytime and from any place in only a few minutes.
Apart from usability and straightforwardness, price is another great thing about DocHub. It has flexible and cost-effective subscription plans and enables you to test our service free of charge during a 30-day trial. Try it out today!
before you take part in this study it is important to fully understand it and to understand what participation may be like researchers will help by providing an informed consent document this is a document that has detailed information about the study including its length the number of visits required medications and the medical procedures in which you will take part the document also provides expected outcomes potential benefits possible risks and other details when needed a translator may be provided researchers will review the informed consent document with you and answer your questions after reviewing the statement getting all the information you need and talking with staff in your family you can decide whether you want to participate if so you will need to sign the informed consent statement your signature indicates that you understand the study and agree to participate voluntarily you may still leave a study at any time and for any reason even after signing the informed consent d