



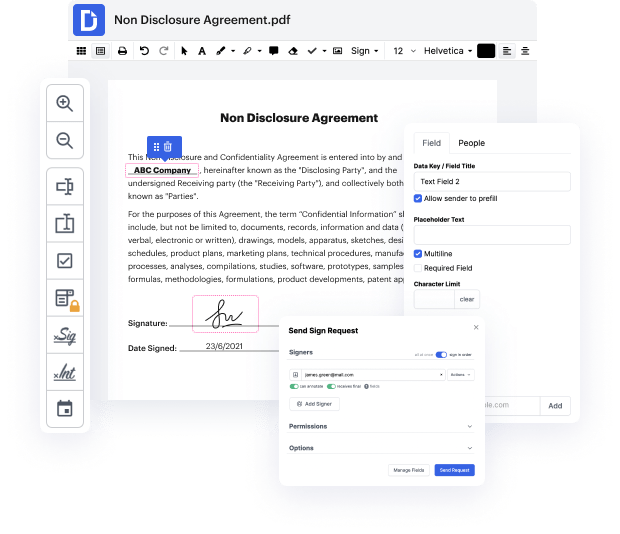
Document generation is a essential aspect of successful organization communication and administration. You require an cost-effective and useful platform regardless of your papers preparation stage. Technology Assessment preparation may be among those procedures which need extra care and attention. Simply stated, there are better possibilities than manually creating documents for your small or medium business. One of the best strategies to ensure good quality and effectiveness of your contracts and agreements is to set up a multifunctional platform like DocHub.
Editing flexibility is regarded as the considerable advantage of DocHub. Employ strong multi-use tools to add and take away, or modify any element of Technology Assessment. Leave comments, highlight information, link sign in Technology Assessment, and transform document administration into an easy and intuitive process. Access your documents at any time and implement new changes anytime you need to, which can considerably reduce your time developing the same document from scratch.
Produce reusable Templates to make simpler your day-to-day routines and get away from copy-pasting the same information repeatedly. Change, add, and adjust them at any moment to make sure you are on the same page with your partners and customers. DocHub helps you avoid errors in frequently-used documents and offers you the very best quality forms. Make certain you always keep things professional and stay on brand with the most used documents.
Enjoy loss-free Technology Assessment modifying and protected document sharing and storage with DocHub. Do not lose any more files or end up perplexed or wrong-footed when discussing agreements and contracts. DocHub empowers specialists everywhere to implement digital transformation as an element of their company’s change administration.


good morning everybody and uh a very warm welcome to this day number three of the training session for patients consumers and Healthcare professionals involved in medicines regulatory activities my name is Michelle Belkin Im heading here at EMA the scientific evidence generation department but Im also coordinating our engagement from EMA side with um other decision makers such as HDA bodies and thats also the particular relationship with todays training which Im super excited about I can tell you its great to have so many participants um here in this meeting we had around 60 registered participants from the patients Healthcare professionals side and this is really also Testament to um this topic being of wider interest and as Maria was just saying um there had been some additional colleagues been added to this session because they hadnt had a chance to hear this before simply because this is the very first time we have HDA Corporation or HDA activities it I should better say u
