



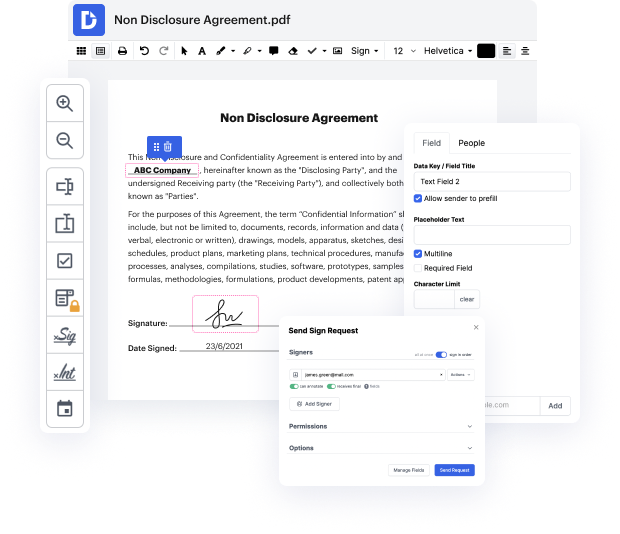
Editing paperwork can be a challenge. Each format comes with its peculiarities, which frequently leads to complex workarounds or reliance on unknown software downloads to bypass them. Luckily, there’s a solution that will make this task less stressful and less risky.
DocHub is a super simple yet full-featured document editing solution. It has a myriad of features that help you shave minutes off the editing process, and the option to Label Comment Letter For Free is only a small part of DocHub’s capabilities.
No matter if if you need occasional editing or to tweak a huge document, our solution can help you Label Comment Letter For Free and make any other desired changes easily. Editing, annotating, certifying and commenting and collaborating on documents is straightforward using DocHub. Our solution is compatible with various file formats - select the one that will make your editing even more frictionless. Try our editor free of charge today!
- Hello everyone, Im Asa Waldstein, and Im here today with some lessons about labeling compliance. Labeling of dietary supplements is very important because if a label format is not correctly, this can really signal to the authorities THAT THERE MIGHT BE DEEPER GMP ISSUES. So this warning letter came from of a company being inspected. During that inspection, the FDA, issued several 483s to the company and they also collected a lot of product labels. So really, I think the reason why this company received the warning letter rather because of their improper 483 response. And I talk more about 483 responses and the differences between a 483 and a warning letter is in a previous post, and Ill drop a link to to that here in the comments. SO THE KEY LEARNING POINTS TODAY have to do with label compliance. SO, LETS REALLY DIG IN ON WHAT WE CAN LEARN. So, a common mistake I see companies making is with supplement fact panels not matching the suggested use. For example, if the suggested use
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



