



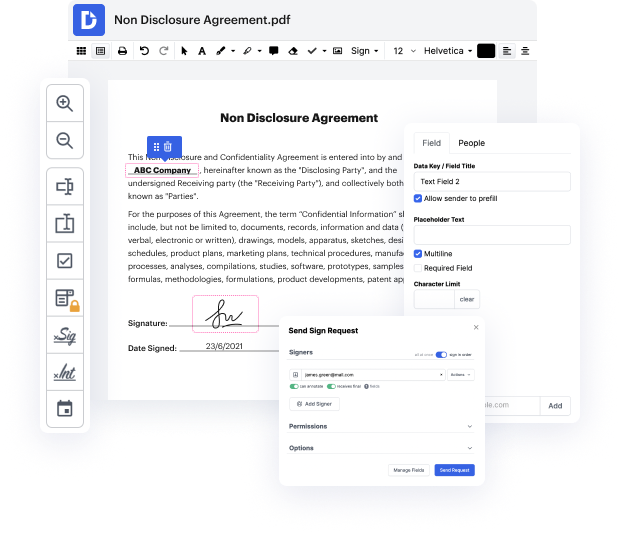
Time is a vital resource that each business treasures and tries to turn into a reward. When choosing document management software, pay attention to a clutterless and user-friendly interface that empowers consumers. DocHub provides cutting-edge tools to maximize your document administration and transforms your PDF file editing into a matter of a single click. Insert List in the Polygraph Consent with DocHub to save a ton of time as well as improve your efficiency.
Make PDF file editing an easy and intuitive operation that saves you a lot of valuable time. Quickly change your documents and send out them for signing without having turning to third-party alternatives. Concentrate on relevant tasks and enhance your document administration with DocHub starting today.
hey everybody welcome to the site owner academy session for this week this week were talking about process of consent and clinical research its one of the most important by far i think in my opinion the most important thing in clinical research that a site will ever do uh and its the process of consent and i dont think theres anything more important than this in research because this informed consent clearly explains the subject that research is experimental and research is not necessarily treatment even though theres treatment aspects to it the research itself is research thats why its called the research and not called treatment right its experimental in nature there are some studies that are more treatment than others but all clinical trials are under the category of research and so this informed consent form is really and the process of consent is really the only way that sites are able to communicate this and document this so so communicate it to the patients and document
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



