



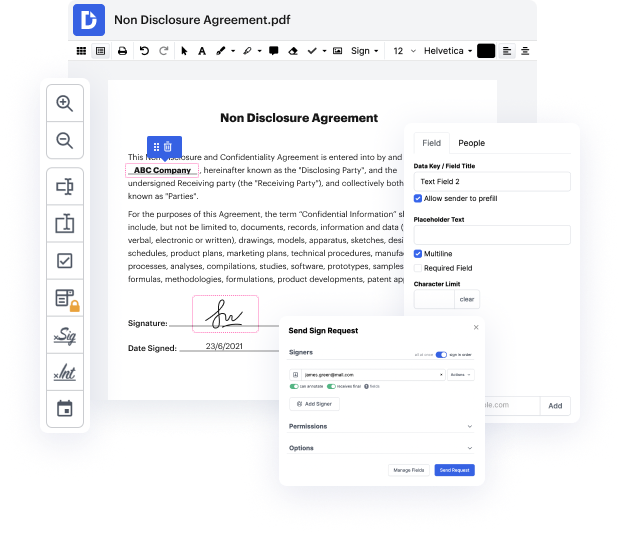
When you want to apply a minor tweak to the document, it should not take long to Inscribe equation notice. This type of simple action does not have to require additional training or running through manuals to understand it. With the proper document modifying instrument, you will not spend more time than is necessary for such a swift change. Use DocHub to simplify your modifying process regardless if you are an experienced user or if it is the first time making use of a web-based editor service. This tool will require minutes or so to learn how to Inscribe equation notice. The sole thing required to get more productive with editing is a DocHub account.
A simple document editor like DocHub can help you optimize the time you need to devote to document modifying no matter your prior knowledge of such instruments. Create an account now and increase your efficiency instantly with DocHub!
in this video were going to talk about how to write net ionic equations so lets consider the reaction between aqueous lead to nitrate and aqueous potassium chloride now the first thing that you need to do is you need to predict the products of this chemical reaction thats the first thing now what we have here is a double replacement reaction lead is going to pair up with chlorine and potassium is going to pair up with nitrate potassium has a positive one charge nitrate has a negative one charge if the magnitude of the charges are the same then you can combine those ions in a one-to-one ratio so one of the products will be kno3 now the other product we need to pair up lead and chloride lead has a positive two charge and we could tell based on how many nitrate ions are attached to it each nitrate ion has a minus one charge and theres two of them so to neutralize the two nitrate ions lead has to have a positive two charge chloride has a minus one charge so when you combine these two
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



