



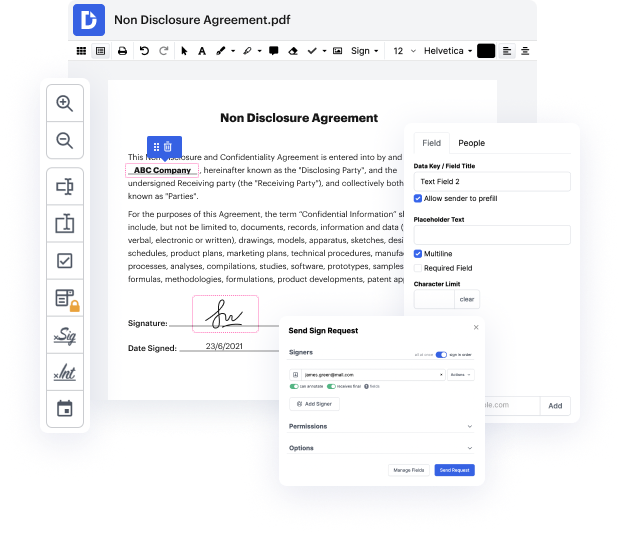
Picking out the excellent document managing platform for the business could be time-consuming. You must assess all nuances of the platform you are interested in, compare price plans, and stay aware with protection standards. Arguably, the ability to work with all formats, including dot, is essential in considering a solution. DocHub offers an extensive list of functions and tools to successfully manage tasks of any difficulty and take care of dot formatting. Register a DocHub account, set up your workspace, and begin dealing with your files.
DocHub is a comprehensive all-in-one program that permits you to edit your files, eSign them, and create reusable Templates for the most commonly used forms. It offers an intuitive interface and the ability to manage your contracts and agreements in dot formatting in a simplified mode. You do not need to bother about reading numerous guides and feeling anxious because the app is way too complex. include formula in dot, assign fillable fields to specified recipients and collect signatures quickly. DocHub is all about powerful functions for experts of all backgrounds and needs.
Enhance your document generation and approval operations with DocHub today. Benefit from all this by using a free trial and upgrade your account when you are ready. Edit your files, create forms, and find out everything you can do with DocHub.
one of the things we do with lewis dot symbols is use them to help us predict the way two elements will come together and bond and thats what were going to be working on on this worksheet so first question we want to predict the compound thats formed between potassium and fluorine to do that were going to begin by drawing the lewis dot symbols of each potassium is in the first column on the periodic table which means in its lewis dot symbol it just has one electron one valence electron and fluorine is in the seventh column on the periodic table which means its lewis dot symbol has seven dots for those seven valence electrons now when these two atoms or any two atoms come together to form a compound they have this overall goal of either creating a situation where they have a total of eight electrons around them in their lewis dot symbol or they have no electrons around them at all so the goal here either is to get like a perfect set of eight we call that an octet or to just strip a
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



