



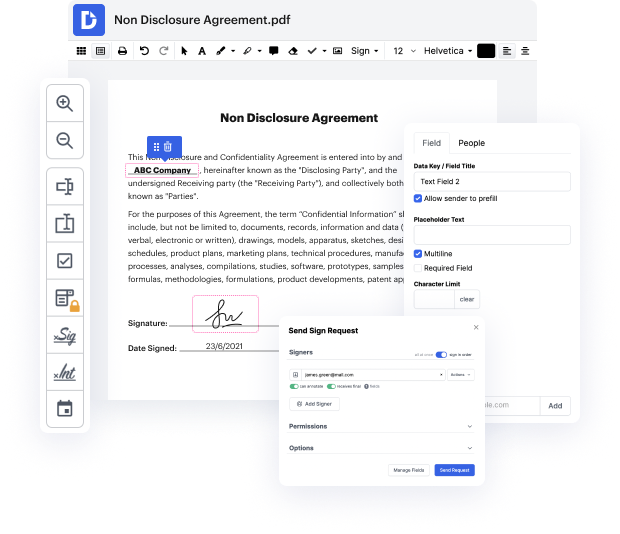
Today’s document management market is enormous, so locating a suitable solution meeting your requirements and your price-quality expectations can take time and effort. There’s no need to waste time browsing the web looking for a universal yet easy-to-use editor to Faint formula in TXT file. DocHub is here at your disposal whenever you need it.
DocHub is a globally-recognized online document editor trusted by millions. It can fulfill almost any user’s request and meets all necessary security and compliance certifications to guarantee your data is safe while modifying your TXT file. Considering its rich and user-friendly interface offered at a reasonable price, DocHub is one of the most beneficial choices out there for enhanced document management.
DocHub offers many other features for efficient form editing. For instance, you can convert your form into a multi-use template after editing or create a template from scratch. Explore all of DocHub’s features now!
I want to show you how we calculate the pH of a solution made using a weak acid in questions involving weak acids not only do you need the concentration of acid in the solution but you also need ka or the acid dissociation constant the KA tells you how much of the acid when its in solution actually dissociates to give you those protons that you need for a solution to be acidic higher values of ka make for more acidic solutions strong acids in fact have the highest Kas because they dissociate completely now when I say higher Kas I want you to be careful 3.4 times 10 to the minus 4 is larger than 3.4 times 10 to the minus 6 when these exponents on the 10 are negative negative 10 to the minus 6 is much smaller than 10 to the minus 4 just be careful of that but a typical question could ask you to calculate the pH of a point 2 molar solution of hydrofluoric acid and they will give you the KA now first thing you have to do is set up this equation for an acid that only gives you 1 H ka repre
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



