



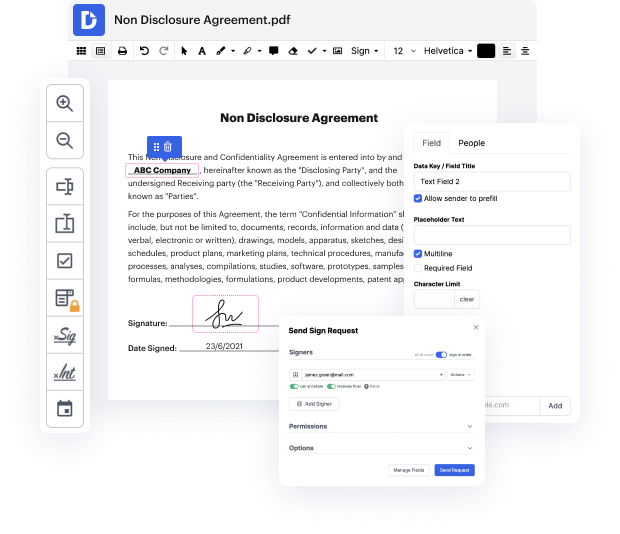
You know you are using the right file editor when such a basic task as Extend date attestation does not take more time than it should. Editing files is now a part of a lot of working processes in numerous professional areas, which is the reason convenience and simplicity are crucial for editing resources. If you find yourself studying tutorials or trying to find tips on how to Extend date attestation, you may want to find a more user-friendly solution to save time on theoretical learning. And this is where DocHub shines. No training is required. Just open the editor, which will guide you through its main functions and features.
A workflow becomes smoother with DocHub. Take advantage of this instrument to complete the documents you need in short time and take your productivity to another level!
this is your GMP minute by Kevin wall today we will discuss how to extend the API expiration date according to IC h q7 drug substances mostly use retest States rather than expiration dates during development expiration dates may not have been determined ongoing stability studies collect data to establish the overall shelf-life an expiry date is a line in the sand beyond which the material cannot be used data demonstrates the amount of time the material is suitable for use retest dates at the time beyond which material should be retested to demonstrate fitness for use both expiry and retest States have supporting stability data as a result development relies on retest dates at the retest date all applicable specifications are verified and the material shown to be conforming the question becomes how long is the use time before the material must be retested again the proper way to extend the retest date is to plot the applicable data examine the plot for linearity for staple materials li
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



