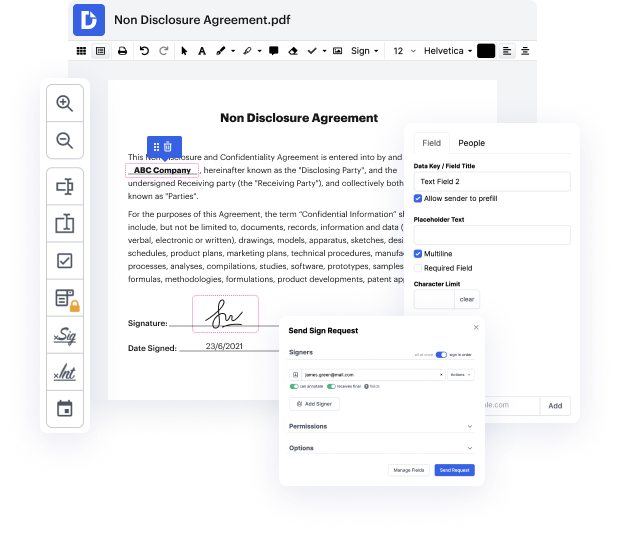
Searching for a professional tool that deals with particular formats can be time-consuming. Regardless of the vast number of online editors available, not all of them are suitable for WRI format, and definitely not all allow you to make changes to your files. To make matters worse, not all of them provide the security you need to protect your devices and paperwork. DocHub is an excellent solution to these challenges.
DocHub is a popular online solution that covers all of your document editing needs and safeguards your work with enterprise-level data protection. It supports different formats, including WRI, and helps you edit such documents quickly and easily with a rich and intuitive interface. Our tool complies with important security certifications, like GDPR, CCPA, PCI DSS, and Google Security Assessment, and keeps enhancing its compliance to guarantee the best user experience. With everything it offers, DocHub is the most trustworthy way to Enter TIN in WRI file and manage all of your individual and business paperwork, regardless of how sensitive it is.
Once you complete all of your modifications, you can set a password on your edited WRI to make sure that only authorized recipients can open it. You can also save your paperwork with a detailed Audit Trail to find out who applied what edits and at what time. Choose DocHub for any paperwork that you need to edit securely. Sign up now!
to write the formula for tin for oxide we look up 10 on the periodic table and the element symbol is s n we then look up oxide thats the oxygen were looking for and thats o tin thats a metal its a transition metal oxygen thats a non-metal so a metal in a non-metal thats an ionic compound we need to think about the charge on the tin and the oxygen when we write the formula it tells us right here tin four that four means the tin has a four plus ionic charge oxygen from the periodic table is two minus since this is a neutral compound we want these to add up to zero to have a net charge of zero and we can see that we need to have two oxygen atoms thatll give us a minus four to balance out the four plus so we could put a subscript right here and now lets check our charges two oxygen atoms each ones two minus so two times two minus thats 4 minus and then we have the 1 4 plus for the 10 so those balance out to 0 net charge is 0. were done this is the formula for 10 4 oxide lets c