



COVID forever changed how firms view their internal protocols and procedures. It affected organizations of all sizes and industries, posing new obstacles for staying connected. The pandemic showed that all firms need to integrate digital instruments into daily routines. They became vital for far more than hybrid working models.
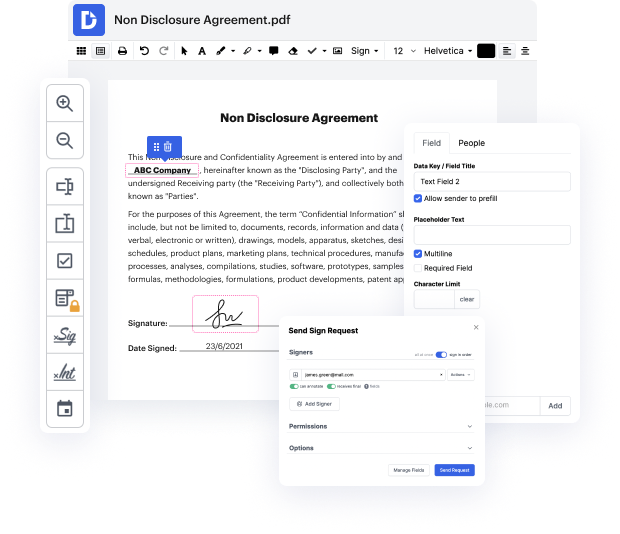
Platforms like DocHub make it easier to increase your document administration and approval operations. DocHub is the go-to tool for end-to-end online editing and signatures. It helps in reducing your day-to-day contract and agreement generation and approval tasks. Obtain access to Document Tool for Medical Device Manufacturers | Medical Device Manufacturers Document Management Solution sophisticated editing features that cover all your administration needs. Deal with any document type and formatting, generate fillable fields, and successfully gather signatures from your teammates and clients. No previous training or experience is required.
With Document Tool for Medical Device Manufacturers | Medical Device Manufacturers Document Management Solution, you can optimize the quality of your files, boost the approval process, and safely store complete files. Obtain a free DocHub profile today and change your subscription when you want.
This video tutorial provides an overview of the documentation required for a medical device development project. The speaker, Peter Sibelius, founder of medicaladvicehq.com, will walk you through how everything fits together, emphasizing the importance of clear view and understanding. The video is part of an online course on design control for medical devices. Bear in mind that this is a simplified version of the real process, and your situation may vary. Subscribe for new content and notifications. Stay tuned for a clear perspective on document deliverables.
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



