



COVID forever changed how organizations look at their internal protocols and processes. It impacted organizations of all sizes and industries, posing new obstacles for staying connected. The pandemic showed that all organizations must integrate digital tools into day-to-day routines. They became important for far more than hybrid working models.
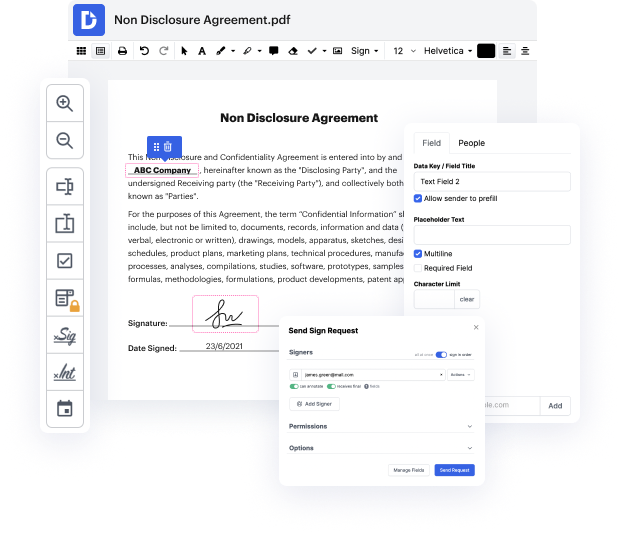
Apps like DocHub allow you to improve your file administration and approval procedures. DocHub is your go-to tool for end-to-end online editing and signatures. It helps reduce your everyday contract and agreement generation and approval tasks. Gain access to Document App for Medical Device Manufacturers | Medical Device Manufacturers Document Management Solution superior editing tools which cover all of your management demands. Deal with any file type and format, create fillable fields, and successfully collect signatures from your teammates and clients. No previous training or experience is required.
With Document App for Medical Device Manufacturers | Medical Device Manufacturers Document Management Solution, you are able to optimize the quality of your documents, accelerate the approval process, and securely store complete documents. Obtain a cost-free DocHub account today and change your subscription when you want.
In this video tutorial, Peter Sibelius, the founder of medicaladvicehq.com, provides an overview of the documentation required for medical device product development projects. He walks through how everything fits together and emphasizes the importance of understanding the big picture in project development. The goal is to provide viewers with a clear view of the process and highlight typical document deliverables. Sibelius encourages viewers to subscribe for future content and offers a disclaimer that individual situations may vary.
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



