



Keeping pace with the fast-changing work environment which has emerged since COVID continues to be an important issue for a lot of industries. Many companies seek out an available and easy-to-adopt platform that will be available for them 24/7. The main interest is based on addressing all document generation and approval operations with minimal effort and time. DocHub presents powerful features and storage space integrations that will boost your every day document operations forever. You simply need a free DocHub user profile to gain access to Doc Editor for Life Sciences | Life Sciences Document Management Solution.
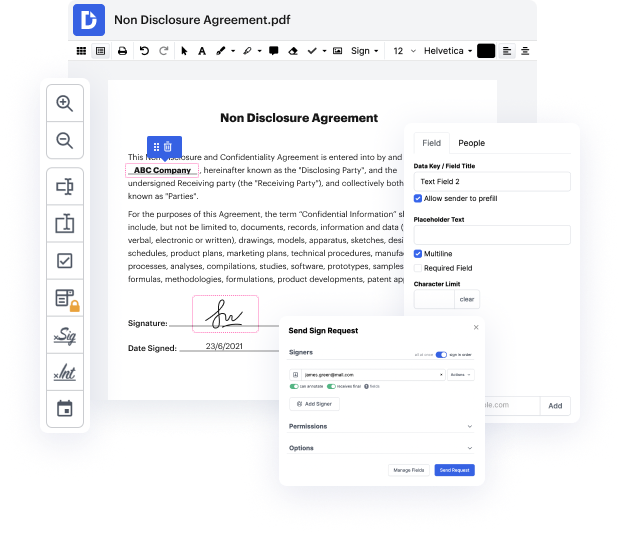
DocHub is an end-to-end editing and annotation solution that handles all your document generation demands. Make documents from scratch, adjust them, leave comments, and quickly gather signatures from your teammates and clients. Forget about losing your documents or worrying about safety - DocHub has industry-leading safety standards that shield your data.
Focus on agreements and contracts anywhere, at any time. Get the most out of Doc Editor for Life Sciences | Life Sciences Document Management Solution and increase your every day document management, from document creation to approval and storage space.
Large complex distributed organizations, especially in the Life Sciences sector, struggle with the proliferation of documents across file shares, SharePoint sites, and in print. Without a clear understanding of what documents exist, their versions, ownership, and control, organizations face challenges. Defined document management processes are crucial for managing policies, procedures, and other critical documents, ensuring ownership, version control, and access. This is essential not only for operational success but also for compliance, risk management, and organizational exposure. Establishing an audit trail of document access is also necessary for accountability and security.
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



