



COVID forever transformed how businesses view their internal practices and processes. It affected companies of all sizes and industries, posing new obstacles for staying connected. The pandemic demonstrated that all businesses must incorporate digital tools into day-to-day routines. They became essential for far more than hybrid working models.
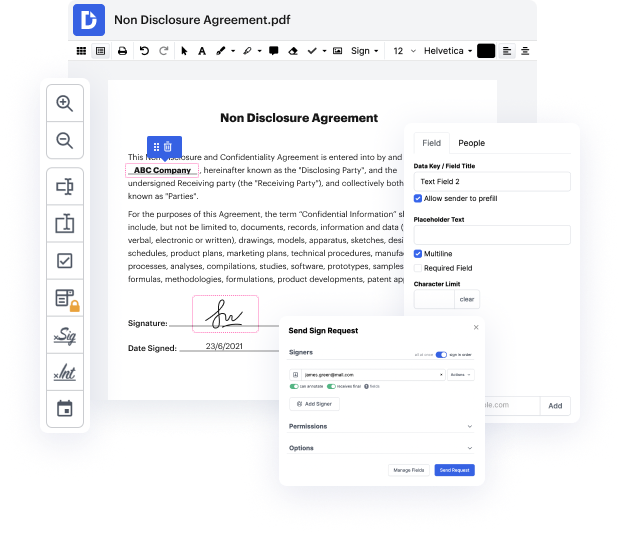
Platforms like DocHub make it easier to enhance your document management and approval processes. DocHub is your go-to instrument for end-to-end online editing and signatures. It helps reduce your daily contract and agreement generation and approval tasks. Get access to Doc Application for Medical Device Manufacturers | Medical Device Manufacturers Document Management Solution superior editing tools that cover all of your managing requires. Work with any document type and formatting, create fillable fields, and effectively gather signatures from your teammates and customers. No previous training or experience is needed.
With Doc Application for Medical Device Manufacturers | Medical Device Manufacturers Document Management Solution, it is possible to maximize the quality of your files, boost the approval process, and safely store finished files. Get yourself a free DocHub profile today and change your subscription when ready.
Creating a testing plan for a startup is crucial and should be done early in the process. Whether you do the pre-sub or not, the same amount will be charged as skipping the pre-sub may lead to more work later. One important part of the pre-sub is covering the testing plan, which includes software validation, EMC testing, electrical safety, and standards such as IEC 62304. This template guides clients through specific questions to ensure thorough testing coverage.
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



