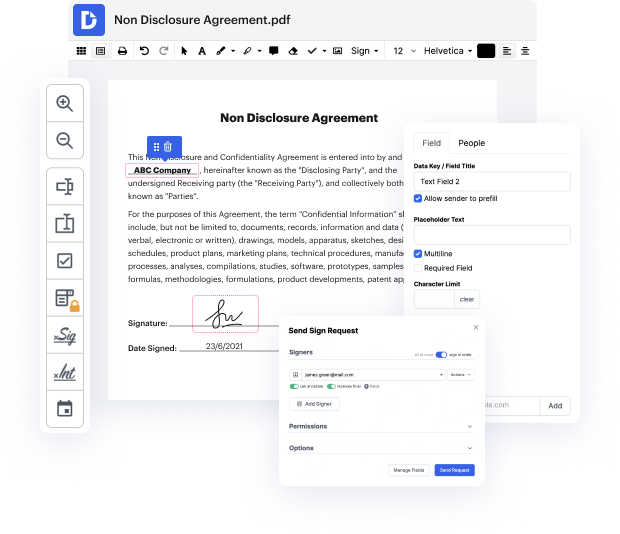
Time is a crucial resource that every enterprise treasures and attempts to change into a benefit. When choosing document management software, take note of a clutterless and user-friendly interface that empowers users. DocHub delivers cutting-edge features to improve your file management and transforms your PDF editing into a matter of one click. Delete Cross Out Option in the Clinical Trial Agreement with DocHub in order to save a ton of time as well as increase your efficiency.
Make PDF editing an easy and intuitive operation that saves you plenty of precious time. Effortlessly change your documents and give them for signing without having turning to third-party software. Give attention to pertinent tasks and improve your file management with DocHub today.
hey everyone its dance fair from the clinical trials guru back with yet another video every day I try to do more than one a day but today Im doing this one its a pretty special video somewhat important as I tell you guys all the time you especially you research clinics but this doesnt just apply for to research clinics it applies to everybody in this industry whether you work for a CRO whether youre a p.i wanting to network with other p is whether your site director of a research clinic even sponsors its so important in this industry to network with your colleagues even if its from competing companies even the Big Pharma is doing this now youre seeing a lot of collaboration going on between Big Pharma so theres no reason as research sites should not be able to do this so Im part of of several groups on LinkedIn there are some Google Plus groups theres my special cyber dusters for those of you who dont know cyber dust is an app you can add me dance vera im planning to start