Document generation and approval are key elements of your daily workflows. These operations are usually repetitive and time-consuming, which effects your teams and departments. Specifically, Clinical Trial Agreement Template creation, storage, and location are important to ensure your company’s efficiency. A thorough online platform can take care of several critical issues related to your teams' efficiency and document management: it takes away tiresome tasks, simplifies the process of locating files and collecting signatures, and leads to a lot more accurate reporting and analytics. That’s when you might require a robust and multi-functional platform like DocHub to handle these tasks swiftly and foolproof.
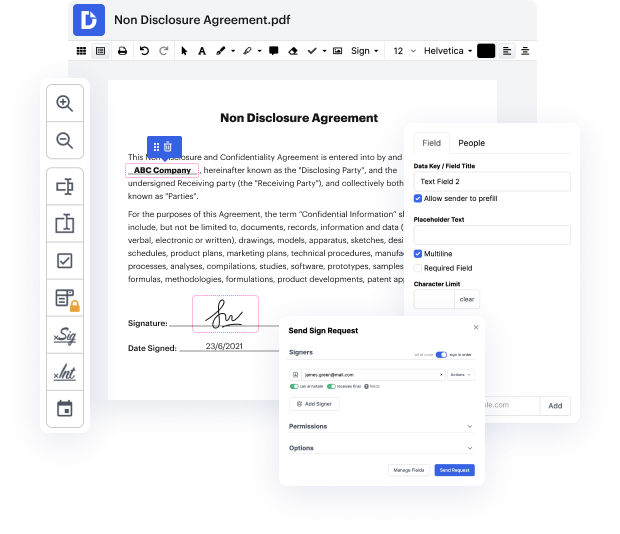
DocHub enables you to streamline even your most complicated process using its powerful functions and functionalities. A strong PDF editor and eSignature transform your everyday document management and turn it into a matter of several clicks. With DocHub, you will not need to look for additional third-party solutions to complete your document generation and approval cycle. A user-friendly interface allows you to begin working with Clinical Trial Agreement Template immediately.
DocHub is more than simply an online PDF editor and eSignature software. It is a platform that assists you easily simplify your document workflows and integrate them with popular cloud storage platforms like Google Drive or Dropbox. Try out editing and enhancing Clinical Trial Agreement Template immediately and discover DocHub's extensive set of functions and functionalities.
Start off your free DocHub trial right now, with no invisible charges and zero commitment. Discover all functions and options of smooth document management done properly. Complete Clinical Trial Agreement Template, gather signatures, and increase your workflows in your smartphone application or desktop version without breaking a sweat. Increase all your daily tasks with the best platform accessible out there.
[Music] this is a demonstration of how to use the practical guidance life sciences practice area to prepare agreements related to the many facets of clinical research for drugs and medical devices one of the fastest and easiest ways to find tools for drafting clinical research agreements is through the clinical trials task on the practical guidance life sciences practice area page here you will find content focused on clinical trials you can get information about fundamentals including contract research organizations irbs and other matters for example this practice note provides drafting tips for contract research organization agreements it also includes a link to cro and research services agreements precedents that will help you to benchmark your agreements with those used by public companies through transaction search by intelligize whether you are tasked with drafting a clinical research support agreement a master clinical trial agreement a material transfer agreement or a sponsore