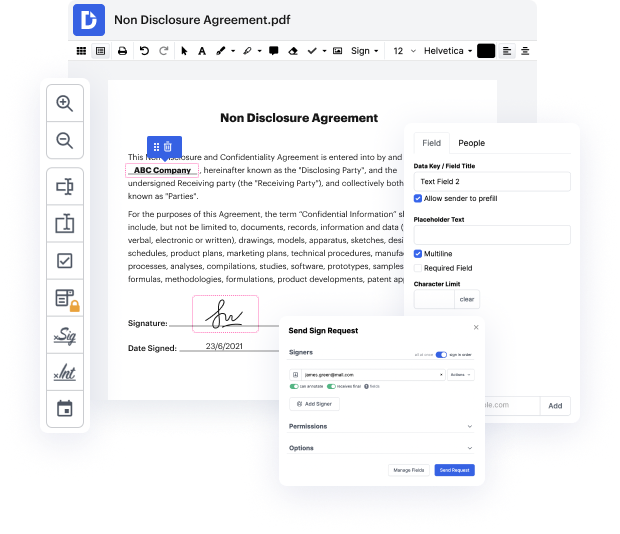
Document generation and approval are core components of your day-to-day workflows. These procedures are frequently repetitive and time-consuming, which influences your teams and departments. Particularly, Video Release Consent Letter creation, storage, and location are significant to ensure your company’s efficiency. An extensive online solution can solve numerous essential issues related to your teams' productivity and document management: it eliminates cumbersome tasks, simplifies the process of finding files and gathering signatures, and results in much more exact reporting and statistics. That is when you may need a robust and multi-functional solution like DocHub to deal with these tasks swiftly and foolproof.
DocHub allows you to simplify even your most complicated task using its strong capabilities and functionalities. A powerful PDF editor and eSignature enhance your day-to-day document administration and make it the matter of several clicks. With DocHub, you won’t need to look for further third-party solutions to finish your document generation and approval cycle. A user-friendly interface lets you start working with Video Release Consent Letter instantly.
DocHub is more than just an online PDF editor and eSignature solution. It is a platform that assists you streamline your document workflows and incorporate them with popular cloud storage platforms like Google Drive or Dropbox. Try editing Video Release Consent Letter immediately and discover DocHub's extensive set of capabilities and functionalities.
Begin your free DocHub trial right now, with no hidden charges and zero commitment. Discover all capabilities and options of smooth document administration done efficiently. Complete Video Release Consent Letter, acquire signatures, and increase your workflows in your smartphone application or desktop version without breaking a sweat. Boost all your day-to-day tasks using the best solution accessible out there.
welcome to the institutional review board for the social and behavioral sciences research education talk on writing an informed consent form at the irb a common question we get from researchers is how do you write a consent form thats why today were going to go over the requirements from the federal regulations and give you tips on writing a consent form the united states department of health and human services has regulations for the protection of human subjects in research the complete regulations can be found at 45 cfr part 46 section 116 but were going to break down and simplify the regulations regarding informed consent here the initial part of the regulations includes a list of concepts as general requirements for informed consent that the researchers should keep in mind and these concepts can be divided into two groups general requirements for informed consent and general requirements for the consent process the first group of general requirements falls under concepts that t