



Working with documents can be a challenge. Each format has its peculiarities, which frequently leads to complex workarounds or reliance on unknown software downloads to avoid them. Luckily, there’s a tool that will make this task more enjoyable and less risky.
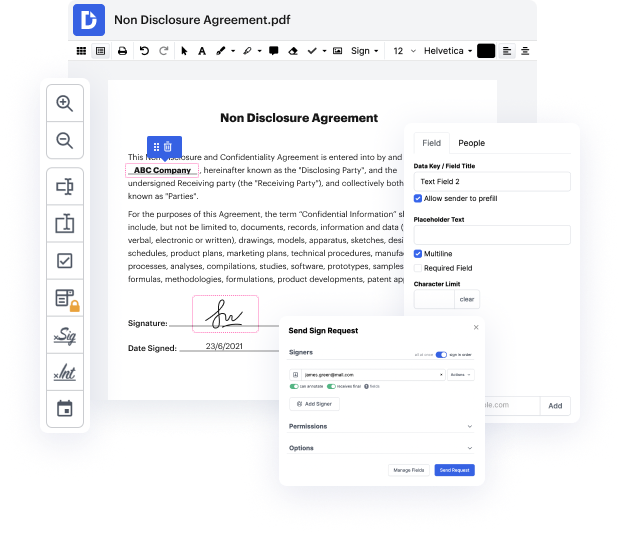
DocHub is a super straightforward yet full-featured document editing program. It has different tools that help you shave minutes off the editing process, and the option to Conform Signature Title For Free is only a small part of DocHub’s functionality.
Whether if you need occasional editing or to tweak a multi-page form, our solution can help you Conform Signature Title For Free and make any other desired changes quickly. Editing, annotating, certifying and commenting and collaborating on files is easy using DocHub. We support different file formats - choose the one that will make your editing even more frictionless. Try our editor free of charge today!
21 CFR part 11 specifies requirements for electronic records and signatures to be equivalent to paper records and handwritten signatures. This allows for the replacement of paper documentation with electronic systems while complying with FDA regulations. The focus of this video is on the requirements for signing documents electronically, with system validation and audit trails covered in another video. Primary requirements include ensuring unique username and password usage, verifying user identity, and notifying FDA when implementing electronic signatures. Users must use two components (username and password) when signing records with electronic signatures.
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



