Working with paperwork implies making minor corrections to them day-to-day. At times, the task runs nearly automatically, especially when it is part of your day-to-day routine. Nevertheless, in other instances, dealing with an uncommon document like a Video Release Consent Letter may take valuable working time just to carry out the research. To make sure that every operation with your paperwork is effortless and fast, you need to find an optimal modifying solution for this kind of tasks.
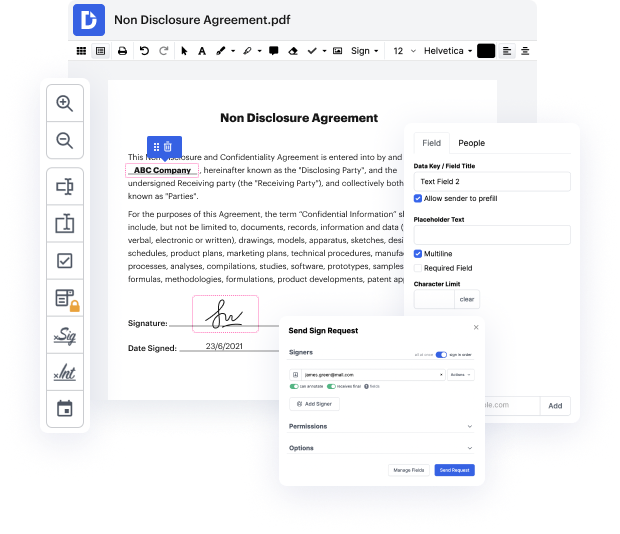
With DocHub, you can see how it works without taking time to figure everything out. Your instruments are organized before your eyes and are readily available. This online solution will not need any sort of background - training or experience - from the customers. It is ready for work even when you are not familiar with software typically utilized to produce Video Release Consent Letter. Easily make, edit, and share documents, whether you work with them daily or are opening a brand new document type for the first time. It takes moments to find a way to work with Video Release Consent Letter.
With DocHub, there is no need to research different document kinds to learn how to edit them. Have all the essential tools for modifying paperwork on hand to streamline your document management.
welcome to the institutional review board for the social and behavioral sciences research education talk on writing an informed consent form at the irb a common question we get from researchers is how do you write a consent form thats why today were going to go over the requirements from the federal regulations and give you tips on writing a consent form the united states department of health and human services has regulations for the protection of human subjects in research the complete regulations can be found at 45 cfr part 46 section 116 but were going to break down and simplify the regulations regarding informed consent here the initial part of the regulations includes a list of concepts as general requirements for informed consent that the researchers should keep in mind and these concepts can be divided into two groups general requirements for informed consent and general requirements for the consent process the first group of general requirements falls under concepts that t